
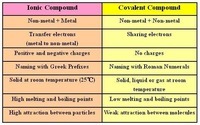
Naming Ionic and Covalent Compounds
Flashcard
•
Other Sciences
•
KG - University
•
Practice Problem
•
Hard

Wayground Content
FREE Resource
Student preview

8 questions
Show all answers
1.
FLASHCARD QUESTION
Front
An ionic compound of calcium and chlorine would be named
Back
calcium chloride.
2.
FLASHCARD QUESTION
Front
A covalent compound made of one sulfur and two oxygen atoms would be named
Back
sulfur dioxide.
3.
FLASHCARD QUESTION
Front
An ionic compound made of copper (Cu2+) and oxygen would be named
Back
copper(II) oxide.
4.
FLASHCARD QUESTION
Front
The covalent compound N2O5 would be named
Back
dinitrogen pentoxide.
5.
FLASHCARD QUESTION
Front
Because the overall charge in a compound must be ____________, the charge of iron in Fe2O3 can be calculated as 3+.
Back
3+
6.
FLASHCARD QUESTION
Front
In the compound TiO2, titanium has a charge of
Back
4+
7.
FLASHCARD QUESTION
Front
The chemical formula for an ionic compound of aluminum and chlorine is
Back
AlCl3.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
