

Valence Bonding Theory
Presentation
•
Chemistry
•
11th Grade - University
•
Medium
Standards-aligned
Jessica Iseman
Used 8+ times
FREE Resource
15 Slides • 11 Questions
1
Valence Bonding Theory

2
Multiple Choice
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice

6

7
Multiple Choice

8

9

10

11

12

13

14

15

16

17

18

19

20

21
Multiple Choice

22
Multiple Choice

23
Multiple Choice

24
Multiple Choice
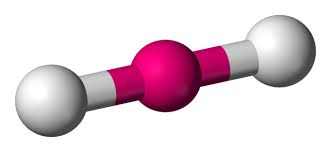
What is the hybridization of a linear molecule?
sp
sp2
sp3
sp3d
25
Multiple Choice
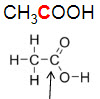
What is the hybridization of the Carbon atom indicated by the arrow?
sp hybridization
sp2 hybridization
sp3 hybridization
dsp3 hybridization
26
Multiple Choice
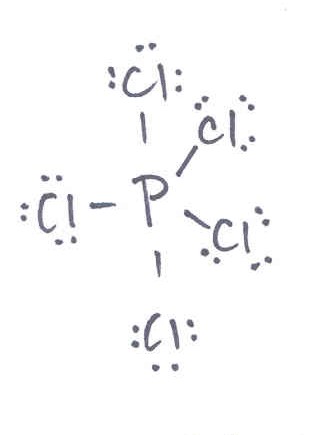
Valence Bonding Theory

Show answer
Auto Play
Slide 1 / 26
SLIDE
Similar Resources on Wayground

20 questions
Leila Ayala
Lesson
•
University

17 questions
M3 L 34
Lesson
•
11th Grade - University

20 questions
Electron Configuration
Lesson
•
9th - 12th Grade

19 questions
Structure of Atom
Lesson
•
9th - 12th Grade

21 questions
Periodic Trends Day One
Lesson
•
10th - 12th Grade

17 questions
Draw Resonance Structures
Lesson
•
10th - 12th Grade

16 questions
Strength of Forces Between Particles
Lesson
•
11th - 12th Grade

20 questions
lesson #28
Lesson
•
12th Grade
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

15 questions
Hargrett House Quiz: Community & Service
Quiz
•
5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

15 questions
Equivalent Fractions
Quiz
•
4th Grade
Discover more resources for Chemistry

30 questions
Element compound mixture review quiz
Quiz
•
10th - 12th Grade

20 questions
U4 Reaction Types
Quiz
•
10th - 12th Grade

25 questions
Potential Energy Diagram Checkpoint
Quiz
•
11th Grade

15 questions
IP PreAP: Solubility Rules and Precipitation Reactions
Quiz
•
University

46 questions
Quantum, E Config, Periodic Trends
Quiz
•
11th Grade

5 questions
DOL REC: Solubility Rules and Precipitation Reactions
Quiz
•
10th - 12th Grade

87 questions
Test 3
Quiz
•
9th - 12th Grade

7 questions
Determine Molecular Formula from Empirical
Quiz
•
9th - 12th Grade