

Science First Semester Study Guide
Presentation
•
Science
•
6th Grade
•
Easy
Standards-aligned
Dawn Kirk
Used 4+ times
FREE Resource
15 Slides • 64 Questions
1
Science First Semester Study Guide
Hello Grade 6 Kirk. View and study the video and other media presentations below. Answer the questions that follow.
Questions are samples of what may come on your science exam. You should also study your notes in your notebook.
Feel free to turn off the music and click the zoom button to make images larger.

2
Topics for Study
Science Tools
Physical Properties
Physical Changes
Chemical Properties and Changes
3
4

5

6
Multiple Choice

List three important pieces of safety gear you need in a lab.
latex gloves, googles, lab coat
gloves, boots, safety gear
7
Multiple Choice

The electronic balance is
used to measure volume of a substance.
mass of a substance or chemical.
used to measure density of a substance or chemical.
8
Multiple Choice

We use a thermometer to measure temperature in ...
degrees Celsius
degrees Fahrenheit
millimetres
degrees Centipede
9
Multiple Choice

What is this piece of laboratory equipment called?
A stand
A tripod
A metal gauze
A rack
10
Multiple Choice

I would use this piece of equipment to ...
find the weight of a substance.
to find the volume of a liquid.
to find the mass of a substance.
to see if I have lost weight.
11
Multiple Select

Check all proper uses for this piece of equipment.
Measuring precise and accurate volume amounts
Heating Solutions
Transferring Solutions
Mixing Solutions
Identifying Mass
12
Fill in the Blanks

Type answer...
13
Multiple Choice
What is a graduated cylinder used for?
Measuring volume
Measuring weight
Pouring liquids
Heating substances
Measuring temperature
14
Multiple Choice

What is this piece of lab equipment called?
wash bottle
stir rod
dropper
funnel
test tube
15
Multiple Choice

this equipment name is . . . .
crucibles
mortars
evaporating dishes
spot plates
16
Multiple Choice

What is the function of an Erlenmeyer Flask?
Used to measure volume very precisely.
A narrow-mouthed container used to transport, heat or store substances; often used when a stopper is required.
A small glass container used to view chemical reactions or to hold or heat small amounts of a substance.
A wide-mouthed container used to transport, heat, or store substances.
17
Multiple Choice
Identify the Erlenmeyer Flask
18
Multiple Choice

What's the name of this lab tool?
Beral Pipet
Scoopula
Stirring Rod
Graduated Cylinder
19
Multiple Choice

What is the function of a beaker?
Used to hold substances being weighed
A small glass container used to view chemical reactions or to hold or heat small amounts of a substance.
A wide-mouthed container used to transport, heat, or store substances.
A narrow-mouthed container used to transport, heat or store substances; often used when a stopper is required.
20
Multiple Choice
Identify the electronic balance.
21
Multiple Choice

Name the equipment
thermometer
dropper pipette
burette
stirring rod
22
Multiple Choice

Name the equipment
graduated cylinder
test tube
thermometer
beaker
23
Multiple Choice

erlenmeyer flask
test tube
graduated cylinder
beaker
24
Multiple Choice

What is the volume of this prism?
16 cubic cm
36 cubic cm
144 cubic cm
25
Multiple Choice

Where should you read the level of water on the graduated cylinder?
Level "A"
Level "B"
Level "C"
None of these
26
Multiple Choice
Volume is the amount of space an object takes up.
True
False
27
Multiple Choice

What is the volume of the water?
24 mL
21.5 mL
20.5 mL
22 mL
28
Multiple Choice

Where do we read the level of liquid?
The highest point the liquid touches.
The bottom of the meniscus.
Half way between, we make an educated guess and round it.
Somewhere about there.
29
Multiple Choice

What is the volume of the liquid in the picture
35 mL
35 grams
35 cm3
35 L
30
Multiple Choice
31
32
Open Ended
What is matter?
33
Open Ended
What is volume?
34
35
Open Ended
What is matter made of?
36
Open Ended
Explain how particles are packed in each of the following:
a. solid
b. liquid
c. gas
37
Multiple Select
True/ False: A solid can become a liquid
False
True
38
39
Multiple Choice

Density of gold
Intensive property
Extensive property
40
Multiple Choice

Boiling point of water
Intensive property
Extensive property
41
Multiple Choice
What kind of physical property is this? Odor
intensive
extensive
42
43
Multiple Choice
What kind of physical property is this? Hardness
intensive
extensive
44
Multiple Choice
What kind of physical property is this? Temperature
intensive
extensive
45
Types of Physical Properties:
Intensive vs. Extensive Properties of Matter
Intensive Properties: color, density, hardness, luster, temperature
Extensive Properties: volume, length, mass weight
Remember: Intensive properties does not depend on the amount of matter present. Extensive properties do depend on the amount of matter present.
46
Multiple Choice
Which set of properties, intensive or extensive, are based on the size of a sample?
intensive
extensive
47
Multiple Choice
Which of the following is an intensive property?
Color
Mass
Volume
Weight
48
Multiple Choice
The mass of a substance divided by its volume.
Volume
Density
Mass
Length
49
Multiple Choice
How shiny a substance is.
Luster
Conductivity
Hardness
Volume
50
Multiple Choice
Which of the following is an extensive property?
Hardness
Boiling Point
Density
Weight
51
Multiple Choice
Which of the following is an extensive property?
Color
Mass
Odor
Luster
52
Multiple Choice
Properties that DO depend on the amount of matter present.
Mass
Hardness
Extensive
Intensive
53
54
Multiple Choice

What is the volume of the liquid in this graduated cylinder?
19 mL
35 mL
24 mL
1.9 mL
55
Multiple Choice

What is the volume of the ring? (new volume - old volume)
64 mL
68 mL
8 mL
4 mL
56
57
Multiple Choice

What is the volume?
length=2 units
width=2 units
height=3 units
Volume = 12 cubic units
Volume = 7 cubic units
Volume = 223 cubic units
Volume = 8 cubic units
58
Multiple Choice

What is the volume?
23 cm3
12 cm3
60 cm3
59
Multiple Choice

Find the volume of the solid shown.
64 in3
21.333 in3
16 in3
50.265 in3
60
Multiple Choice

61
62
63
Multiple Choice
Which of the following indicates a chemical change?
Change in state of matter
Temperature change
Change in size
Change in shape
64
Multiple Choice
Evidence of a physical change.
New substance
Shape Change
Light produced
Gas released.
65
Multiple Choice
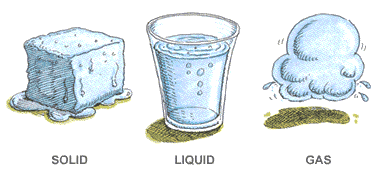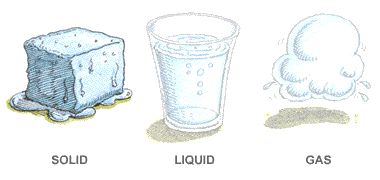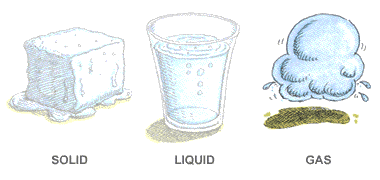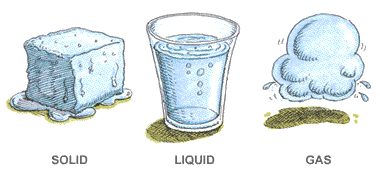
In which of these do particles move slowest?
matter
gas
liquid
solid
66
Multiple Choice

Is this an example of a physical or chemical change?
Physical because it is a mixture.
Chemical because the parts have changed shape.
67
Multiple Choice

Wood that is burning in a brush pile has heat, and produces ashes.
Physical
Chemical
68
Multiple Choice

Is this an example of a physical or chemical change?
physical
chemical
69
Multiple Choice

Is this an example of a physical or chemical change?
physical
chemical
70
Multiple Choice
A chemical change
changes the physical properties of an object, but does not create a new substance.
creates a new substance and cannot be undone.
71
Multiple Choice
A physical change...
changes the physical properties of an object, but does not create a new substance.
creates a new substance and cannot be undone.
72
73
Multiple Choice

Molecules change position easily and slide past one another.
Solid
Liquid
Gas
74
Multiple Choice
Matter that has no definite shape, nor volume.
Solid
Liquid
Gas
75
Multiple Choice
What happens when my glass of water is sitting in the sun and disappears into a gas?
evaporation
burning
melting
freezing
76
Multiple Choice

The boiling point of water in oCelsius and oFahrenheit is:
0oC or 32oF
100oC or 212oF
WRONG
WRONG
77
Multiple Choice

When a gas changes into a liquid, it is called ......
evaporation
condensation
melting
freezing
78
Multiple Choice

When a solid changes into a liquid, it is called ......
freezing
sublimation
melting
boiling
79
Multiple Choice

The freezing point of water in oCelsius and oFahrenheit is:
100oC or 212oF
0oC or 32oF
WRONG
WRONG
Science First Semester Study Guide
Hello Grade 6 Kirk. View and study the video and other media presentations below. Answer the questions that follow.
Questions are samples of what may come on your science exam. You should also study your notes in your notebook.
Feel free to turn off the music and click the zoom button to make images larger.

Show answer
Auto Play
Slide 1 / 79
SLIDE
Similar Resources on Wayground

74 questions
TLT Ch. 10
Presentation
•
6th Grade

76 questions
Human Reproductive System
Presentation
•
6th Grade

75 questions
European Governments: UK, Germany, and Russia
Presentation
•
6th Grade

71 questions
7.3.1-Solids, Liquids, & Gases
Presentation
•
7th Grade

76 questions
Southwest Asia and North Africa "The Middle East"
Presentation
•
6th Grade

68 questions
CLIMATE REVIEW
Presentation
•
6th Grade

74 questions
Kisi - kisi Ujian Madrasah 2021
Presentation
•
6th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

18 questions
Oceans, Atmosphere, Climate Vocabulary Amplify
Quiz
•
6th Grade

20 questions
Cell Organelles
Quiz
•
6th Grade

25 questions
animals
Quiz
•
6th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade