

ATOMIC STRUCTURE QUIZ
Presentation
•
Science
•
8th Grade
•
Medium
+1
Standards-aligned
CherryAnn Comeros
Used 14+ times
FREE Resource
1 Slide • 13 Questions
1
ATOMIC STRUCTURE QUIZ

2
Multiple Choice

3
Multiple Choice
Valence electrons are the...
Innermost electrons
Middle electrons
Outermost electrons
Any electrons
4
Multiple Choice
5
Multiple Choice

How many valence electrons?
2
4
6
8
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice

9
Multiple Choice

10
Multiple Choice
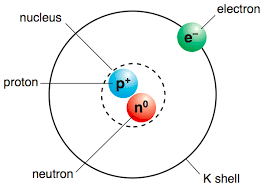
11
Multiple Choice

What subatomic particles would you find in the nucleus of an atom?
Protons only
Protons and Neutrons
Neutrons and Electrons
Protons and Electrons
12
Multiple Choice
Which of these statements is FALSE?
Electrons have a negative charge.
Electrons have a mass of 1 amu.
The nucleus of an atom is positively charged.
The neutron is found in the nucleus of an atom.
13
Multiple Choice
The 2 main parts of an atom are:
nucleus & electron cloud
nucleus & protons
protons & electrons
neutrons & electron cloud
14
Multiple Choice

What subatomic particle(s) would be found orbiting the nucleus?
Neutrons only
Electrons only
Protons and Neutrons
Protons and Electrons
ATOMIC STRUCTURE QUIZ

Show answer
Auto Play
Slide 1 / 14
SLIDE
Similar Resources on Wayground

7 questions
Mendeleev to Modern
Presentation
•
8th Grade

7 questions
Development of the Periodic Table
Presentation
•
8th Grade

7 questions
Table of Elements
Presentation
•
8th Grade

7 questions
Developing the Periodic Table
Presentation
•
8th Grade

10 questions
WhatisEvolution?
Presentation
•
8th Grade

9 questions
Progression of Atomic Models
Presentation
•
8th Grade

15 questions
Cell and cell organelle
Presentation
•
8th Grade

14 questions
Bonding and Parts of a Chemical Equation
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade

20 questions
Energy Transformations
Quiz
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade