

Isotopes in Use
Presentation
•
Science
•
11th - 12th Grade
•
Easy
Standards-aligned
Kristy Hilt
Used 9+ times
FREE Resource
7 Slides • 11 Questions
1

Now that you've found the mass of isotopes on your own, let's see how scientists actually look at isotopes!
Isotopes
2
Isotopes: How are they Measured?
A mass spectrometer is an instrument used to separate an element’s isotopes and to measure the relative isotope abundances.
3
Isotopes: How are they Measured?
How does a spectrometer work?
Within the spectrometer, beams made of charged atoms (ions) from one element are passed through a strong magnetic field. The ions are selected to have identical charges and velocities.
As the ions pass through the magnetic field, the ion paths are influenced by the magnetic force.
Ions of greater mass possess greater inertia or a greater tendency to continue in a straight line. Ions of lesser mass are more influenced by the field and demonstrate greater deviation.
VIDEO TIME!: How does a spectrophotometer work?
4
Isotopes: How are they Measured?
What does a spectrometer produce?

Here is an example of a mass spectrometer reading. It displays the result of passing ions of an element through a spectrometer.
*Note the aces
**The Y-axis should say relative percent abundance
5
Isotopes: How are they Measured?
What does this tell us?

This element has 1 isotope with a mass of 27.
Most elements have more than 1 isotope, resulting in more bars on this type of graph. An example is next!
You will need to determine the element then write each isotope in isotope notation.
6
Isotopes: How are they Measured?
Now you try.
Check it out. Questions coming on the next two slides.
Identify the element.
Isotope Notation.

7
Fill in the Blanks

Type answer...
8
Draw
Represent each isotope in proper isotope notation.
9
Isotopes: How are they Measured?
Another Example.
Check it out. Questions coming on the next two slides.
Identify the element.
Isotope Notation.

10
Fill in the Blanks

Type answer...
11
Draw
Represent each isotope in proper isotope notation.
12
Multiple Select
Select all that is true.
Isotopes of the same atom have...
the same number of protons.
the same mass.
different quantities of neutrons.
different masses.
13
Multiple Choice
How many protons does P-30 have?
14
Multiple Choice
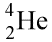
15
Multiple Choice
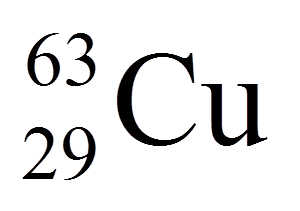
16
Multiple Choice
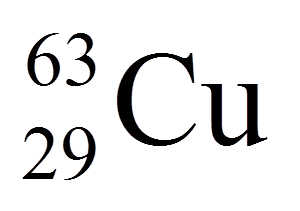
17
Multiple Choice
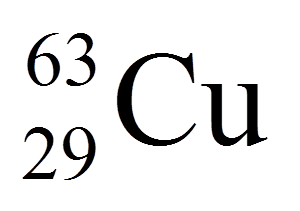
18
Multiple Choice


Now that you've found the mass of isotopes on your own, let's see how scientists actually look at isotopes!
Isotopes
Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

17 questions
Fossil Fuels - Introduction
Presentation
•
11th - 12th Grade

12 questions
Renewable Energy
Presentation
•
11th - 12th Grade

13 questions
Speed v. Velocity
Presentation
•
8th Grade

12 questions
Prepositions
Presentation
•
3rd Grade

15 questions
Transformations of Parent Functions
Presentation
•
11th - 12th Grade

10 questions
Genetic Engineering
Presentation
•
10th - 11th Grade

13 questions
Renewable and Nonrenewable Resources
Presentation
•
12th Grade

14 questions
GTS
Presentation
•
11th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

28 questions
Greenhouse effect
Quiz
•
9th - 12th Grade