

Chemistry Essentials Review
Presentation
•
Science, Chemistry
•
9th - 12th Grade
•
Hard
Connor Boyd
FREE Resource
53 Slides • 17 Questions
1

2


3

4

5


6

7

8

9

10

11


12


13


14


15


16


17


18


19

20

21
Multiple Select
Which particles have approximately the same size and mass?
Protons
Neutrons
Electrons
Atoms
22
Multiple Select

The nucleus of an atom contains which subatomic particles? (Choose 2)
Protons
Neutrons
Electrons
Protons, Neutrons, and Electrons
23
Multiple Choice
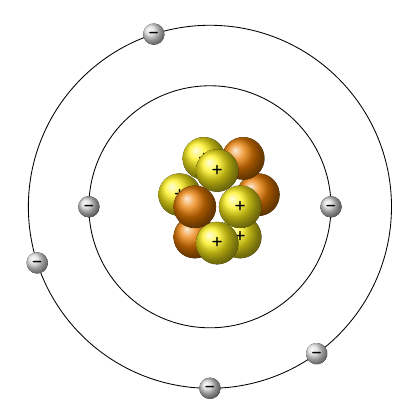
How many electrons does this atom have?
2
4
6
10
24
Multiple Choice
in the nucleus
in the protons
in the neutrons
in the electrons
25
Multiple Choice
How many Subatomic Particles are there in an atom?
1
2
3
26
Atoms, Elements, Molecules, Compounds

27

28

29

30

31

32

33

34
Multiple Choice
Which substance is a compound?
Neon (Ne)
Oxygen (O2)
Carbon Dioxide (CO2)
Nitrogen (N2)
35
Multiple Choice
Which substance is not an element?
Neon (Ne)
Oxygen (O2)
Carbon dioxide (CO2)
Iron (Fe)
36
Multiple Choice
Is Sodium (Na) an element or a compound?
Element
Compound
37
Multiple Choice
Is Iron (Fe) an atom or a molecule?
Atom
Molecule
38
Multiple Choice
Is Chlorine (Cl2) an atom or a molecule?
Atom
Molecule
39

Ionic Bonding
40


Chemical bonding
An attraction between atoms that allows the
formation of substances with two or more atoms.
41


Atom
The smallest unit of matter that forms a chemical
element.
42

Types of chemical bonding
Ionic bonding
Covalent bonding
Metallic bonding
43


Ionic bonding
The transferring of valence electrons to create oppositely
charged ions that are attracted to each other.
The atom that is a metal will always donate to the atom
that is a nonmetal.
44


Valence electrons
Electrons on the outermost energy level.. “Ring”
45


Ions
an atom with a positive or
negative charge due to the
loss or gain of electrons.
46


Ionic bonding
The transferring of valence electrons to create oppositely
charged ions that are attracted to each other.
47



Ionic bonding
Atoms either want to fill or “unload” their outer ring.
“Unload” - Sodium
“Fill” - Chloride
48


Ionic bonding of Sodium Chloride
49

50
Multiple Choice
What is the basis of ionic bonding?
share electrons
transfer electrons
51
Multiple Choice
molecule
ion
isotope
element
52

53

54
Sharing is Caring!
C3.5 - Covalent Bonding

55
Sharing instead of Transferring
Nonmetals share electrons instead of pulling electrons because they have relatively small electronegativity differences and have similar affinities to electrons.
56
Compounds are made of two or more atoms of different elements.
Atoms can be bonded ionically (charge) or covalently (by sharing electrons)
"All compounds are molecules but not all molecules are compounds"

57
Covalent compounds have a MOLECULAR FORMULA.
Shows the types and numbers of atoms in a single molecule of a molecular compound (a covalent substance).
"Molecular Compound" = "Covalent Compound"

58
Covalent bonds form from shared electrons.
Atoms have lower potential energy when bonded (more stability

59
Atoms tend to form bonds to follow the octet rule.
A "full outer shell" means more stability/lower potential energy.
H and He are exceptions to the octet rule.

60
Valence electrons are involved in bonding.
The group number can tell you how many valence electrons an atom has.
Groups 1 and 2 have "1" and "2" e- respectively.
Group 13 has "3",, group 14 has "4", and so on through group 18...

61
Dots are used to represent valence electrons in an electron-dot notation.
Each dot represents an electron.
A total of 4 PAIRS of electrons shows an octet

62
Lewis structures show atoms as they are bonded to each other.
Dots are unpaired electrons
Dashes are a shared pair of electrons
Single bonds = 1 line = 1 PAIR of electrons
Multiple bonds = more than 1 line

63
Multiple Choice
What two types of atoms make a covalent bond?
2 non-metals
1 metal and 1 non-metal
2 metals
64
Multiple Choice
What type of bond involves the sharing of electrons between atoms?
covalent bond
ionic bond
metallic bond
transfer bond
65
Multiple Choice
Identify the following compound as ionic or covalent: SO2
ionic
covalent
66
Bohr Models

67

68
Multiple Choice
What is a valence electron?
Electrons in the first energy shell
Electrons in the second energy shell
Electrons in the outer shell
Total number of electrons
69

70
Multiple Choice

Which element is this?
Nickel
Neon
Sodium
Nitrogen

Show answer
Auto Play
Slide 1 / 70
SLIDE
Similar Resources on Wayground

67 questions
Magnetism and Electromagnetism Review
Presentation
•
11th - 12th Grade

59 questions
Matter and Energy
Presentation
•
9th - 11th Grade

66 questions
High School Organic Chemistry
Presentation
•
9th - 12th Grade

69 questions
Ch. 5 Electrons in Atoms
Presentation
•
9th - 12th Grade

61 questions
Science of Chemistry Mr. Bradley
Presentation
•
9th - 12th Grade

69 questions
SES1a Formation of the Solar System
Presentation
•
9th - 12th Grade

69 questions
Dynamic Equilibrium
Presentation
•
10th - 11th Grade

66 questions
Science Unit 2 of Term 2 (Water)
Presentation
•
KG - Professional Dev...
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

22 questions
School Wide Vocab Group 1 Master
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

10 questions
Exploring the Layers of the Earth
Interactive video
•
6th - 10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

15 questions
Protein synthesis
Quiz
•
9th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Newton's Laws of Motion
Quiz
•
9th Grade

20 questions
Mendelian Genetics Review
Quiz
•
9th Grade

16 questions
Balancing Chemical Equations and Types of Reactions
Quiz
•
9th - 10th Grade