

REVIEW DENSITY GRADE 7
Presentation
•
Science, Physics
•
KG
•
Practice Problem
•
Hard
Noble Teacher
Used 1+ times
FREE Resource
12 Slides • 21 Questions
1

2

3
Multiple Choice
Density is ____
the amount of mass in an object
the amount of mass in a given space
the amount of space an object takes up
the weight of an object
4

5

6
Multiple Choice

7

8

9

10
Multiple Choice

Show all your work on your answer document. D=m/v
0.33 g/cm3
15 g/cm3
3 g/cm3
9 g/cm3
11
Multiple Choice

Show all your work on your answer document. D=m/v
0.5 g/cm3
6 g/cm3
2 g/cm3
4 g/cm3
12
Multiple Choice

Show all your work on your answer document. D=m/v
0.5 g/cm3
8 g/cm3
2 g/cm3
4 g/cm3
13
Multiple Choice
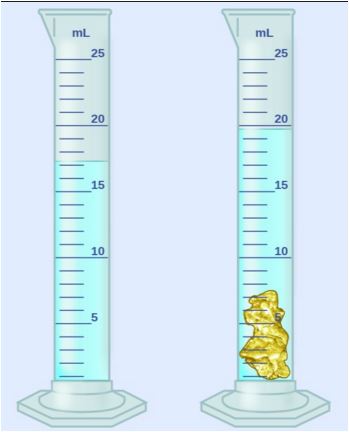
14
Multiple Choice

15
Multiple Choice

16
Multiple Choice
What scientific device would be used to measure the volume of an irregular object?
beaker
scale
graduated cylinder
triple beam balance
17
Multiple Choice
If water has a density of 1 g/cm3, what would an object with a density of 1.5 g/cm3 do in water?
sink
float
I don't know
18

The different densities of objects determines if it will sink or float in water.
Higher density than water will SINK!
Lower density than water will FLOAT!
The density of water is defined as 1 gram / milliliter3
What makes objects sink or float?
19
Multiple Choice
Calculate the density of a rock with the mass of 108 grams and a volume of 12 cm3.
9 g/cm3
0.11111 cm3/g
I have no idea!
19 g/cm3
20
One way would be to put the object in liquid that is inside a graduated cylinder. The liquid in the cylinder will rise the same amount as the volume of the object.
Subtract the final liquid volume reading from the starting volume reading to find the volume of the object.
Science tool definition: A graduated cylinder is a tall narrow cylinder with markings used to measure volume of liquids!
So, then, how do you measure the volume of an irregular shaped object?

21

Rectangular objects - calculate volume by using the formula:
Volume = Length x Width x Height.
Volume is measured in different ways depending on what shape the object has.
Let's practice using this formula on the next slide.
22
Multiple Choice
Calculate the volume of a block that is 5 m long, 6 m wide, and 2 m high.
10 m3
30 m2
60 m3
120 m3
23

All matter takes up space. The amount of space an object takes up is called its volume.
Volume can be measured in cubic meters (m3), cubic centimeters (cm3), liters, and milliliters (mL).
What is volume?
24

Mass is defined as the amount of matter an object has. It is not affected by gravity, and doesn't change based on location.
Mass differs from weight because weight is affected by gravity. Weight is the force of gravity pulling down on you.
Mass is measured in kilograms.
Fun fact! You will weigh less on the moon than you do on Earth! But you will weigh more on Jupiter!
Mass
25
Multiple Choice
Frank has an eraser. It has a mass of 4g, and a volume of 2cm3. What is its density?
8 g/cm3
2 g/cm3
1/2 g/cm3
24 g/cm3
26
Multiple Choice
What is the formula for density?
density = mass x volume
density = mass / volume
density = mass + volume
density = mass - volume
27
Multiple Choice
Jack has a rock. The rock has a mass of 14g and a volume of 2cm3. What is the density of the rock?
7 mL
7 g/cm3
28 g/cm3
1/7 g/cm3
28
Multiple Choice

What is the volume of the rock?
30cm3
10cm3
40cm3
20cm3
29
Multiple Choice

30
Multiple Choice
If a block of wood has a density of 0.6 g/cm3 and a mass of 120 g, what is its volume?
200 cm3
0.005 cm3
200.0 ml
72 ml
31
Multiple Choice

Put the liquids in order from most dense to least dense?
4, 3, 2, 1
1, 2, 3, 4
3, 4, 2, 1,
4, 3, 1, 2
32
Multiple Choice
Frank has a paper clip. It has a mass of 9g and a volume of 3cm3. What is its density?
3 g/cm3
1/3 g/cm3
27 g/cm3
39 g/cm3
33
Multiple Choice
Which units of measurement are used for volume?
grams
centimeters
grams/milliliter
milliliter

Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

26 questions
Mass Extinction
Presentation
•
6th - 8th Grade

26 questions
Space Missions
Presentation
•
8th - 9th Grade

26 questions
SC.5.L.14.2 Comparing Plants and Animals
Presentation
•
3rd - 5th Grade

26 questions
Pure Substances vs. Mixtures
Presentation
•
7th Grade - University

27 questions
Ecosystems
Presentation
•
8th Grade

27 questions
Tropisms Notes
Presentation
•
6th - 8th Grade

26 questions
Volcanoes
Presentation
•
6th Grade

26 questions
Volcanoes
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade