

Solutions 2024 Lesson
Presentation
•
Science
•
12th Grade
•
Easy
+9
Standards-aligned
Candice R. HENRY
Used 7+ times
FREE Resource
16 Slides • 29 Questions
1

2

3
Multiple Choice
What are the phases of matter?
solid
liquid
gas
plasma
solid
water
rock
electricity
solid
liquid
gas
magma
solid
liquid
mixture
pure substance
4
Multiple Choice
An elements is an example of a _________.
pure substance
mixture
homogeneous mixture
heterogeneous mixture
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice

Type of mixture that has the SAME COMPOSITION in every part.
Homogenous
Heterogeneous
11

12

13

14
Multiple Choice

15
Multiple Choice
How can Rey make the soup cubes dissolve more quickly in the water?
16
Multiple Choice
Why is Darla stirring the iced tea?
17
Multiple Choice

Three 10 g samples of sugar are represented below.
Sample A dissolves in water more slowly than sample B.
Sample B dissolves more slowly than sample C.
Which of the following best explains why sample A dissolves more slowly than the other two?
It has the most volume.
It has the smallest surface area.
It has the largest number of sugar molecules.
It has the fewest bonds between sugar modules.
18

19
Multiple Choice

20

21

22

23

24

25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
Which of the following best describes a supersaturated solution?
a solution that contains the maximum amount of solute that is capable of being dissolved
a solution that contains less than the maximum amount of solute that is capable of being dissolved.
a solution that contains more than the maximum amount of solute that is capable of being dissolved.
none of the above
28
Multiple Choice

How does a solution become supersaturated?
Vigorous stirring to dissolve more solute than usual.
Heat the solution to increase the solute then let it cool.
Add more solvent to lower the concentration.
Keep pouring solute in the solvent until it goes in.
29
Multiple Choice
What do you call a solution that contains less than the maximum amount of solute that is capable of being dissolved?
unsaturated solution
saturated solution
supersaturated solution
insoluble solution
30
Multiple Choice
When the solution equilibrium point is reached and no more solute will dissolve, the solution is said to be _________.
unsaturated
saturated
supersaturated
31
Multiple Choice
When a solvent contains as much of a solute as it can hold, it is described as being
saturated
unsaturated
supersaturated
miscible
32

33

34

35
Multiple Choice
Two liquids that can be mixed together but separate shortly after are:
immiscible
insoluble
miscible
soluble
36
Multiple Choice
Oil does not dissolve in water. Which term would best describe a mixture of oil and water?
miscible
immiscible
saturated
unsaturated
37
Multiple Choice
If you increase the temperature of a solution you ________________the solubility of most substances.
increase
decrease
don't alter
38
Multiple Choice
Solubility of gases are greater in ___________________ water.
cold
warm
hot
room temperature
39

40
Multiple Choice
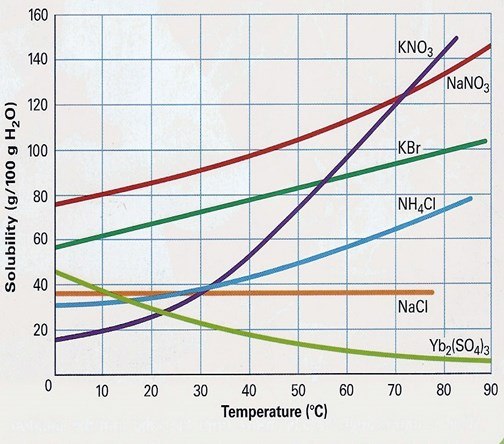
Identify if the following solution would be saturated, unsaturated, or supersaturated: 103 g KBr at 70'C.
41
Multiple Choice
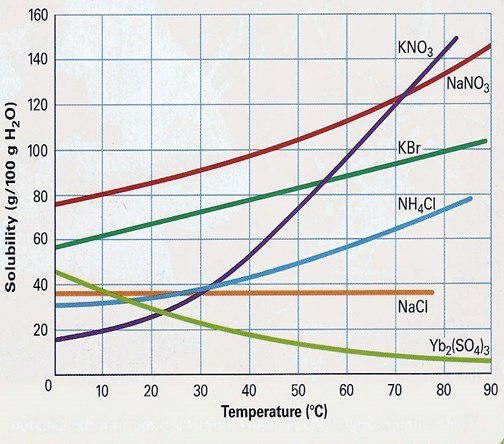
Identify how many grams of KNO3 is required to make a saturated solution at 40'C.
50 g
45 g
60 g
33 g
42
Multiple Choice

What type of a solution is 60g NaNO3 at 40ºC in 100g H2O?
Saturated
Unsaturated
Supersaturated
43
Multiple Choice

What type of a solution is 25g NaCl at 70ºC in 100g H2O?
Saturated
Unsaturated
Supersaturated
44
Multiple Choice

What type of a solution is 55g KCl at 70ºC in 100g H2O?
Saturated
Unsaturated
Supersaturated
45


Show answer
Auto Play
Slide 1 / 45
SLIDE
Similar Resources on Wayground

40 questions
Sources of Energy
Presentation
•
11th - 12th Grade

43 questions
Exercise during pregnancy
Presentation
•
12th Grade

40 questions
Medical Terminology
Presentation
•
12th Grade

40 questions
Unit A - Properties of Energy
Presentation
•
12th Grade

40 questions
Class 12 EVS 2017 Paper
Presentation
•
12th Grade

37 questions
Tragedy Notes
Presentation
•
12th Grade

38 questions
Arthropoda
Presentation
•
11th - 12th Grade

42 questions
IB Diploma Programme Exams
Presentation
•
11th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

28 questions
Greenhouse effect
Quiz
•
9th - 12th Grade