

CHEM Chapter 22 Sec 2
Presentation
•
•
Practice Problem
•
Hard
Bianka Salichs
FREE Resource
5 Slides • 102 Questions
1
Multiple Choice
Is the name of single bonded, saturated hydrocarbons.
Alkanes
Alkenes
Alkynes
Tanins
2
Multiple Choice
Is the name of doubled bonded, unsaturated hydrocarbons.
Alkanes
Alkenes
Allkynes
Tanins
3
Multiple Choice
Is the name of triple bonded, unsaturated hydrocarbons.
Alkanes
Alkenes
Alkynes
Tanins
4
Multiple Choice
Is the first step to name a Hydricarbons
Number the carbons
Name the branches
Find the longest/main chain
Number the branches
5
Multiple Choice
is the alkene with 3 carbons in its chain.
Ethene
Propene
Methene
Decene
6
Multiple Choice
Is the simplest alkane
Ethane
Methane
Propane
Decane
7
Multiple Choice
Is a characteristic of alkyles
Triple Bonded
Double Bonded
Single Bonded
Saturated
8
Multiple Choice
Is the last step in naming alkenes and alkyne.
Writing the name of the main chain.
Writing the location of the double/triple bond and the name of the main chain.
Writing the location of the branches and the name of the main chain.
9
Multiple Choice
Is the simplest of the alkanes.
Methane
Ethane
Propane
Buthane
10
Multiple Choice
Is the simplest of alkenes.
Ethene
Propene
Buthene
Pentene
11
Multiple Choice
Is the simplest of alkynes.
Ethyne
Propyn
Butyn
Pentyne
12
Multiple Choice

Is the name of the representation in the image
Condensed Formula
Line Diagram/Skeletal Formula
Structural Formula
Molecular Formula
13
Multiple Choice

Is the name of the representation showed in the image.
Condensed Formula
Structural Formula
Line Diagram/Skeletal Formula
14
Multiple Choice
Is the name of the nomenclature seen in class.
IUPAC nomenclature
TUPAC nomenclature
ICE-T nomenclature
ZUPAC nomenclature
15
Multiple Choice
Choose the alkene
16
Multiple Choice
Choose the alkane
17
Multiple Choice
Choose the alkyne
18
Prefixes for carbon atoms
Compounds with single bonds are alkanes: -ane
Compounds with double bonds are alkenes: -ene
Compounds with triple bonds are alkynes: -yne

19
Therefore a hydrocarbon with 2 carbons is:
Alkane: ETHANE
Alkene: ETHENE
Alkyne: ETHYNE

20

21
Multiple Choice
What is the name of 4 carbons alkane?
Butane
Butyne
Butene
22
Multiple Choice
What is the name of 11 carbons alkyne?
Undecyne
Undecane
Undecene
23
Multiple Choice

What is the name of this compound?
octane
octane
octyne
24
Start numbering from the end nearer the double bond
The locator for the bond is the lowest number.
Alkenes or alkynes with two or three carbons do not need numbers.
The name of this alkene is 2-heptene or hept-2-ene.

25
The name of this alkyne is ...
3-nonyne or non-3-yne.

26
Multiple Choice

Choose the name of the compound
3-butyne
2-butyne
3-butene
2-butene
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice

1-propene
2-propyne
30
Multiple Choice
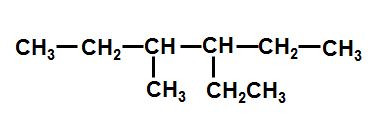
Give the IUPAC name for this compound
3-methyl-4-ethylhexane
3-ethyl-4-methylhexane
3-methyl-4-ethylheptane
3-ethyl-4-methylheptane
31
Multiple Choice
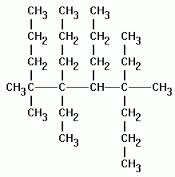
32
Dropdown
33
Categorize







Organize these hydrocarbons into these categories
34
Dropdown
35
Categorize






Organize these isomers into cis or trans isomers
36
Draw
Draw the product below
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
What is another name for carbon compounds?
carbohydrates
fullerenes
hydrocarbons
organic compounds
42
Multiple Choice
43
Multiple Choice
44
Multiple Choice

45
Multiple Choice
46
Multiple Choice
What is the general formula of the alkenes?
CnH2n+1
CnH2n+2
CnH2n
CnH2n+1OH
47
Multiple Choice
What is the suffix for double bond hydrocarbons ?
-ol
-ene
-ane
-one
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
51
Multiple Choice
52
Multiple Choice

53
Multiple Choice

54
Multiple Choice

55
Multiple Choice

56
Multiple Choice

57
Multiple Choice

58
Multiple Choice

59
Multiple Choice

60
Multiple Choice
Which are the following are all alkanes?
methane, ethane, propane
methane, ethene, propene
methene, ethene, propene
ethene, propene, butene
61
Multiple Choice
What term is given to a compound which contains atoms of carbon and hydrogen only?
alkene
hydrocarbon
alkane
saturated
62
Multiple Choice
What term describes alkane molecules that contain C-C single bonds?
saturated
unsaturated
63
Multiple Choice
64
Multiple Choice
Is the chemistry of carbon.
Inorganic Chemistry
Organic Chemistry
Analytical Chemistry
Biochemistry
65
Multiple Select

Which vocabulary words can be used to describe this image? (check all that apply)
molecule
compound
element
atom
66
Multiple Choice

What type of model is pictured here?
space filling model
ball and stick model
67
Multiple Choice

Which of these is only a molecule and not a compound?
a
b
c
d
68
Multiple Choice

What type of model is this?
Dot and cross diagram
Displayed formula
Ball and stick model
Space filling model
69
Multiple Choice

What type of model is this?
Dot and cross diagram
Displayed formula
Ball and stick model
Space filling model
70
Multiple Choice

What type of model is this?
Dot and cross diagram
Displayed formula
Ball and stick model
Space filling model
71
Multiple Choice
Which of the following shows a dot and cross diagram
72
Multiple Choice
Which of the following shows a ball and stick model?
73
Multiple Choice
Which of the following does not give any information about the bonding within the molecule?
C2H6
74
Multiple Choice
Which of the following is good for showing the 3-dimensional arrangement of the atoms?
C2H6
75
Multiple Select
Which of the following is able to show the bonds between atoms?
C2H6
76
Multiple Choice
What prefix do we use for alkanes that are arranged in a loop?
ring-
cyclic-
cyclo-
77
Multiple Choice

Name this molecule
butane
propane
cyclobutane
cyclopentane
78
Multiple Choice
79
Multiple Choice
80
Multiple Choice
81
Multiple Choice
82
Multiple Choice

Name.
4-methylhexane
3-methylhexane
1-ethyl-2-methylbutane
other
83
Multiple Choice

Name.
1, 2-diethyl-3-methylbutane
4-ethylheptane
4-methylseptane
other
84
Multiple Choice

Name.
4-ethyl-3, 3, 4--methylheptane
4-ethyl-4, 5, 5-trimethylheptane
4-ethyl-3, 3, 4--trimethylheptane
other
85
Multiple Choice

Name.
1-ethyl-1, 2, 2, 3-butmethyloctane
4-ethyl-2, 3, 3, 4-tetramethyloctane
1, 3-diethyl-2, 2, 3-trimethylheptane
other
86
Multiple Choice

Name.
3, 4, 5-triethyl-4-methylnonane
3, 4, 5-tridiethyl-4-methylnonane
4-methyl-3, 4, 5-triethylnonane
other
87
Multiple Choice

Name.
4, 5-ethyl-3, 4-methyl-4-propylheptane
4, 5-diethyl-3, 4-dimethyl-4-propylheptane
2, 3, 4-triethyl-3-methyl-4-propylheptane
other
88
Multiple Choice
What is the main component of natural gas?
butane
methane
propane
ethane
89
Multiple Choice
What are some common sources of natural gas?
Shale formations, coal beds, and oil fields
Volcanic eruptions, geothermal reservoirs, and oceanic crust
Natural springs, underground rivers, and glaciers
Wind turbines, solar panels, and hydroelectric dams
90
Multiple Choice
What are some industrial uses of natural gas?
Medical equipment, renewable energy, and entertainment industry
Power generation, heating and cooling systems, manufacturing processes, and as a feedstock for the production of chemicals and fertilizers
Transportation fuel, cooking and baking, and construction materials
Water treatment, telecommunications, and clothing production
91
Multiple Choice
What are some environmental advantages of using natural gas?
Natural gas produces fewer greenhouse gas emissions and air pollutants compared to other fossil fuels.
Natural gas does not contribute to air pollution.
Natural gas is a renewable energy source.
Natural gas has no negative impact on ecosystems.
92
Multiple Choice
What are some economic advantages of using natural gas?
Lower cost, abundant supply, and reduced greenhouse gas emissions.
Higher cost, abundant supply, and increased greenhouse gas emissions.
Higher cost, limited supply, and no impact on greenhouse gas emissions.
Higher cost, limited supply, and increased greenhouse gas emissions.
93
Multiple Choice
What are some safety concerns associated with natural gas?
Risk of leaks, explosions, fires, and carbon monoxide poisoning
Environmental impact and greenhouse gas emissions
Limited availability in certain areas
High cost of installation and maintenance
94
Multiple Choice
What are some potential environmental disadvantages of using natural gas?
Increased energy efficiency, reduced carbon emissions, and improved air quality
Decreased reliance on fossil fuels, lower electricity costs, and reduced water usage
Enhanced energy security, reduced dependence on foreign oil, and increased job creation
Greenhouse gas emissions, air pollution, and water pollution
95
Multiple Choice
Which of the following is a fossil fuel?
Petroleum
Wood
Cow dung
Dry leaves
96
Multiple Choice
Identify the fuel that leaves more smoke and ash on burning.
Solid fuel
Liquid fuels
Gaseous fuels
Any one of the above
97
Multiple Choice
98
Multiple Choice
99
Multiple Choice
100
Multiple Choice
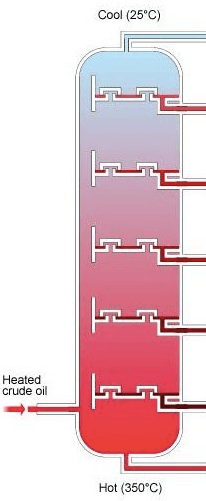
What process happens as the crude oil enters the fractionating column?
Evaporation
Condensation
Combustion
101
Multiple Choice
These statements about aromatic compounds are correct EXCEPT
They are cyclic and planar
They follow Huckel's 4n rule
They have conjugated double bonds
They have delocalized undefinedelectrons
102
Multiple Choice
Which of the following molecules is aromatic?
103
Multiple Choice
Benzene can only undergoes reaction of ______________
elimination
addition
substitution
none
104
Multiple Select
TRUE OR FALSE:
Aromatic compounds are good solvents for nonpolar solute.
TRUE
FALSE
105
Multiple Choice

Because they have aromas, compounds with a ring of resonance bonds are called ___
fragrent compounds
pungent molecules
aromatic compounds
scented structures
106
Multiple Choice

Is this aromatic compound or not?
Yes
No
107
Multiple Choice
Which compound below is aromatic?
Is the name of single bonded, saturated hydrocarbons.
Alkanes
Alkenes
Alkynes
Tanins
Show answer
Auto Play
Slide 1 / 107
MULTIPLE CHOICE
Similar Resources on Wayground

101 questions
Chapter 13- Medical Math
Presentation
•
9th - 12th Grade

101 questions
El Pretérito 2023
Presentation
•
10th - 12th Grade

102 questions
F5 (L) Module L29
Presentation
•
10th Grade

103 questions
Evolution EOC Review
Presentation
•
9th - 12th Grade

101 questions
WAVES
Presentation
•
9th - 12th Grade

103 questions
network standards
Presentation
•
9th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade