

3.6 Pressure in iquids and gases
Presentation
•
Physics
•
7th Grade
•
Practice Problem
•
Hard
+2
Standards-aligned
Jake Villagonzalo
FREE Resource
20 Slides • 43 Questions
1

Pressure in
liquids and gases
MR. JAKE
2

LEARNING OBJECTIVES
Here’s what you’ll learn in this lesson:
Explain that pressure is caused by the action of a force, exerted by a substance, on an area (pressure=force/ area)
Use particle theory to explain pressure in gases and liquids
3

Success Criteria
I understand what causes pressure in a liquid.
I understand how pressure changes with depth in a liquid.
I understand what causes pressure in a gas.
I understand how altitude affects atmospheric pressure.
I understand how the quantity of gas in a container affects the pressure.
I understand how the temperature of a gas in a closed container affects the pressure.
4

Watch this video to explore more about pressure in liquids. It is important that you pause the video and take down notes.
5
Multiple Choice
In which directions do liquids exert pressure within a container?
Only on the base
Only on the walls
On the base and walls
Liquids do not exert pressure
6
Multiple Choice
Which layer of water in a container experiences the greatest pressure?
Top layer
Middle layer
Bottom-most layer
All layers have equal pressure
7
Multiple Choice
When making a hole near the top layer of water in a container, how does the water flow out?
Straight down
At an angle
Along the walls
Does not flow out Answer
8
Multiple Choice
Why does water squirt out more forcefully from holes near the bottom of a container?
Higher water pressure at the bottom
Lower water pressure at the bottom
Equal pressure throughout
Pressure depends only on container size
9

Pressure and
Depth in
Liquids
10


Pressure in Water
Divers have to be careful if they wantto dive to a great depth in the sea.
The weight of the water above presses down on them. The greater
the depth they go, the more water
there is above them pressing down.
So, what conclusion can you make about the pressure of water?
11

Pressure and depth in liquids
The pressure in a liquid increases with depth,
but why?
As you go deeper in a liquid, there is more liquid above you. The weight of this liquid, caused by gravity, pushes on the particles of the liquid.

12

Pressure and depth in liquids
When the particles of the liquid are pushed, the force on the particles increases.
As the particles in a liquid are moving randomly in all directions, then the pressure in the liquid is equal in all directions.

13


Atmospheric pressure is greatest at sea level. This is because, at sea level, we have the weight of the whole atmosphere above us, pressing down. It is as though we live at the bottom of an “ocean air”.
14


As the diver dives deep down to the depth of the ocean/water, the pressure increases because the particles of water pushes or the weight of the water presses on the diver.
15
Open Ended
Using the concept of pressure in liquids, explain why does the pressure increases as the diver dives deeper into the ocean or into the water?
16
Multiple Choice
Why does pressure increase as you go deeper in a liquid?
The particles become smaller
There is more liquid above exerting weight
Gravity decreases with depth
Liquid density decreases
17
Multiple Choice
What role does gravity play in the pressure of a liquid at greater depths?
It reduces pressure
It pushes liquid particles upward
It causes the weight of the liquid above to increase pressure
It only affects the surface
18
Multiple Choice
How do liquid particles contribute to pressure at a specific depth?
They move randomly in all directions
They only move downward
They are stationary
They repel each other
19
Multiple Choice
In which directions is pressure equal at a given depth in a liquid?
Only upwards
Only downwards
In all directions
Only sideways
20
Multiple Choice
Why do the particles in a liquid contribute to pressure in all directions?
Because they are all stationary
Because they move randomly
Due to the force of gravity alone
They only move downwar
21
Multiple Choice
Which of the following best explains why liquid pressure is different from atmospheric pressure?
Liquid particles are stationary while gas particles move freely
Liquid pressure depends on depth, while atmospheric pressure depends on altitude
Liquid pressure depends on temperature
Liquid pressure is unaffected by depth
22
Multiple Choice
If a diver goes 10 meters below the surface of a lake, why does the pressure on them increase?
The diver’s weight increases at greater depths
More water above adds weight, increasing pressure
Gravity is stronger underwater
Water density decreases with depth
23
Multiple Choice
Which factor directly affects the pressure on a submerged object in a liquid?
The object’s mass
The surface area of the object
The depth of the object in the liquid
The object's shape
24
Multiple Choice
If a submarine descends deeper into the ocean, why does the pressure on it increase?
The ocean temperature increases
More water above the submarine exerts weight
The submarine’s density decreases
Ocean currents apply more force at depth
25
Multiple Choice
Why does pressure increase on the walls of a container as more water is added?
The container expands, increasing pressure
The added water exerts greater weight downward and outward
Water molecules become more compact
The temperature of the water rises
26
Multiple Choice
When a diver goes from 5 meters to 15 meters underwater, why does the pressure on them change?
The diver becomes denser
The water pressure from above increases with more depth
The diver's temperature affects surrounding pressure
Water density decreases at greater depths
27

Pressure in
Gas
28

Before you blow up a balloon, there is a small volume of air inside the balloon. The balloon is open at one end, so the pressure of the air inside is the same as the pressure of the air outside

29

As you blow air into the balloon, you are adding more gas particles. Particles in a gas move randomly and collide with the walls of the container.


Every time a gas particle collides with the wall of the container, the particle exerts a small force on the wall. The more particles there are in the gas, the more collisions happen with the walls, and so the force on the walls increases. As this force is exerted on an area, the force causes pressure. The pressure inside the balloon gets bigger as you blow in more air, pushing the walls of the balloon outwards
30
Multiple Choice
Before you blow up a balloon, why is the air pressure inside it the same as the outside?
Because no air particles are inside
The balloon is open, equalizing pressure with outside air
The balloon has a vacuum inside
The balloon walls exert outward pressure
31
Multiple Choice
What happens to the amount of gas particles inside the balloon as you blow into it?
It decreases
It remains the same
It increases
It fluctuates
32
Multiple Choice
How do gas particles move within a container?
In a fixed pattern
In straight lines only
Randomly
Downward only
33
Multiple Choice
How does adding more gas particles affect the frequency of particle-wall collisions in the balloon?
Increases collision frequency
Decreases collision frequency
Has no effect on collisions
Makes collisions random
34
Multiple Choice

How is pressure within the balloon created?
By the balloon material
By the collisions of gas particles with the walls
By air outside the balloon
By gravity pulling down on the balloon
35
Multiple Choice
Why does atmospheric pressure vary at different altitudes?
Different altitudes have different gravitational forces
The concentration and weight of air particles change with altitude
Humidity increases with altitude
Temperature decreases with altitude
36

As you go higher in the atmosphere, two variables that affect atmospheric pressure change:
• the number of particles in 1 m3 of air decreases, so the concentration of gas particles decreases
• the weight of air above your current position decreases.
Pressure and depth
in Gas
Atmospheric pressure at sea level is about 100 000 N/m2 .
This pressure is the equivalent of the weight of two large elephants pushing on every 1m2 of surface!
We are not aware of the pressure from the atmosphere because we live in the atmosphere all the time.
37



Pressure in Gas
Why do climbers of the Mt.Everest have to wear a gas mask at the summit?
38
Open Ended
Why do climbers of the Mt.Everest have to wear a gas mask at the summit?
39


If you climb a high mountain, the air gets thinner (less dense) as you go higher. Its density decreases. The air is less dense at the top of a mountain, its pressure is less. We call the pressure of the air atmospheric pressure.
Pressure of the
Atmosphere
40
Multiple Choice

As altitude increases, what happens to the number of gas particles in 1 m³ of air?
(altitude means the height or distance above sea level)
It increases
It decreases
It stays the same
It fluctuates randomly
41
Multiple Choice
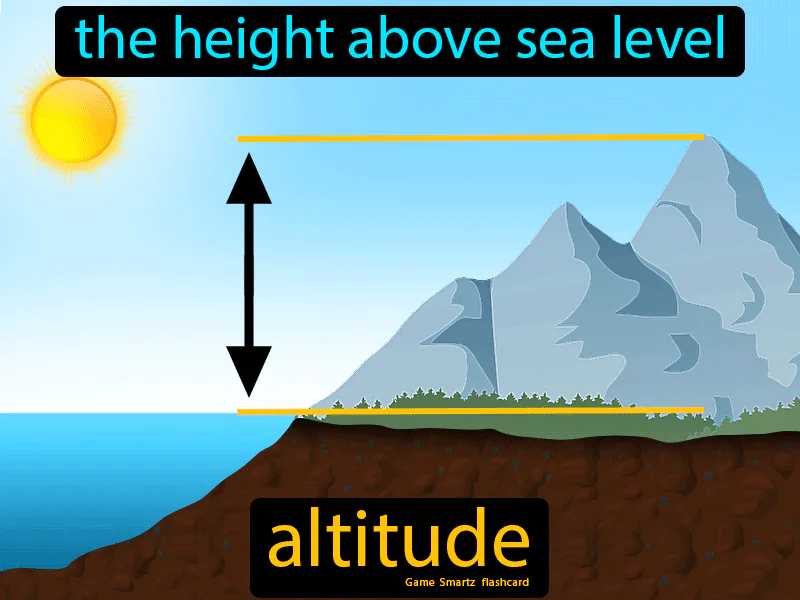
Which of the following statements is true about atmospheric pressure as altitude increases?
It increases with altitude
It decreases with altitude
It remains constant
It depends on humidityIt depends on humidity
42
Multiple Choice
Why does atmospheric pressure decrease at higher altitudes?
Air density increases with altitude
There are fewer gas particles and less weight of air above
Gravity is stronger at higher altitudes
Temperature is higher at higher altitudes
43
Multiple Choice
What is the approximate atmospheric pressure at sea level?
50,000 N/m²
75,000 N/m²
100,000 N/m²
150,000 N/m²
44
Multiple Choice
Atmospheric pressure at sea level is roughly equivalent to the weight of:
One large elephant on every square meter
Two large elephants on every square meter
Ten small birds on every square meter
A small dog on every square meter
45
Multiple Choice
Why are humans generally unaware of atmospheric pressure?
The atmosphere exerts no pressure on humans
We are used to it as we live in the atmosphere constantly
It only affects objects at high altitudes
The human body resists atmospheric pressure
46
Multiple Choice
How does the weight of the air above change as altitude increases?
It increases
It decreases
It stays constant
It depends on temperature
47
Multiple Choice
What happens to the concentration of gas particles in the atmosphere with altitude?
It increases
It decreases
It remains the same
It varies randomly
48
Multiple Choice
Which force is responsible for the pressure exerted by the atmosphere at sea level?
The weight of the air above
The temperature of the atmosphere
The speed of wind currents
The rotation of the Earth
49
Multiple Choice
Why does atmospheric pressure vary at different altitudes?
Different altitudes have different gravitational forces
The concentration and weight of air particles change with altitude
Humidity increases with altitude
Temperature decreases with altitude
50

The effect of atmospheric pressure can be shown by pumping the air out of a metal container.
Before the air is pumped out, the pressure on the inside of the container is equal to the pressure on the outside.
Effects of atmospheric pressure
When the air is pumped out, the pressure inside the container becomes close to zero.
The pressure on the outside does not change. The container is crushed by the pressure of the air outside the container. The picture shows a conttainer crushed by atmospheric pressure.
51
Multiple Choice
Before the air is pumped out of the container, why does it not get crushed?
The pressure outside is higher than inside
The pressure inside equals the pressure outside
The container is very strong
Atmospheric pressure only affects open containers
52
Multiple Choice
What happens to the pressure inside the container when air is pumped out?
It increases slightly
It decreases close to zero
It remains the same
It becomes higher than outside pressure
53
Multiple Choice
Why does the container get crushed after the air is pumped out?
Internal pressure increases
Outside pressure exceeds the pressure inside
Air rushes back into the container
The container has no structural support
54
Multiple Choice
When the air is pumped out, what does the remaining atmospheric pressure do to the container?
It stretches it
It compresses it
It cools it
It causes it to expand
55
Multiple Choice
Which concept does the crushed container best demonstrate?
The effects of reduced internal pressure
The force of gravity
How gases expand when heated
Conservation of energy
56

As the temperature of a gas increases, the speed of the particles in the gas increases. When particles are travelling faster, their collisions exert more force. This means that "increasing the temperature of a gas will increase the pressure"
of the gas.
Pressure and Temperature in Gases
57
Multiple Choice
What happens to the pressure in a sealed container of gas if the temperature increases?
Pressure decreases
Pressure increases
Pressure stays the same
Pressure fluctuates randomly
58
Multiple Choice
If you heat a gas in a balloon, what will likely happen to the balloon’s size?
The balloon will expand
The balloon will shrink
The balloon will remain the same size
The balloon will deflate
59
Multiple Choice
Why does increasing the temperature of a gas increase its pressure?
Particles slow down, exerting more force
Particles move faster, colliding more forcefully
Particles stay the same speed but grow in size
The volume of the container increases
60
Multiple Choice
In a closed metal container, what would happen to gas pressure if the container is left in the sun?
The pressure will decrease as it cools
The pressure will increase as it heats
Pressure won’t change
Pressure decreases initially, then increases
61
Multiple Choice
If a gas’s temperature is doubled in a fixed-volume container, what effect does this have on particle collisions?
Collisions decrease in frequency
Collisions become weaker
Collisions become stronger and more frequent
Collisions remain constant
62

Success Criteria
I understand what causes pressure in a liquid.
I understand how pressure changes with depth in a liquid.
I understand what causes pressure in a gas.
I understand how altitude affects atmospheric pressure.
I understand how the quantity of gas in a container affects the pressure.
I understand how the temperature of a gas in a closed container affects the pressure.
63

Do you have any questions?
v.jake2@vinschool.edu.vn
Thanks!

Pressure in
liquids and gases
MR. JAKE
Show answer
Auto Play
Slide 1 / 63
SLIDE
Similar Resources on Wayground

58 questions
Untitled Presentation
Presentation
•
7th Grade

60 questions
Dividing Integers
Presentation
•
7th Grade

55 questions
Meiosis
Presentation
•
7th Grade

56 questions
Simple and Extended Structures
Presentation
•
7th Grade

57 questions
Periodic Table Quiz Lesson Review
Presentation
•
7th Grade

59 questions
S1_5.4/5.7 Characteristics: TRF & Mangroves (Notes)
Presentation
•
7th Grade

57 questions
Indus Valley Civilization
Presentation
•
6th - 7th Grade

61 questions
Rock Cycle (lesson)
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade