

Transfer of Energy
Presentation
•
Science
•
6th Grade
•
Medium
+6
Standards-aligned
NaaJah Richardson
Used 7+ times
FREE Resource
14 Slides • 12 Questions
1
Transfer of Energy
8.SC.15 Use models to construct an explanation of how energy is transformed but still conserved.
By NaaJah Richardson
2
Matter
State of Matter
Temperature
Volume
Mechanical Energy
Energy Transfer
Environment
Factors
Kinetic Energy
Mass
Academic Vocabulary
3

Energy is the ability to do work or cause change (moving, heating, or changing the state of matter).
Energy
4

Energy cannot be created or destroyed, only transferred or transformed.
Example: A roller coaster moving from potential to kinetic energy.
Image: Roller coaster energy transformation diagram
Law of Conservation of Energy
5

It is the sum of potential energy and kinetic energy of the entire system.
ALL Forms of energy are measured in joules (J).
Mechanical Energy
6
Work
Work is the transfer of energy to an object by a force that makes the object move in the direction of the force.
Ex. You pulled back on the pendulum with a force. So, you did work on the pendulum.
7
Multiple Choice

At which point is gravitational potential energy at its maximum?
8
Multiple Choice
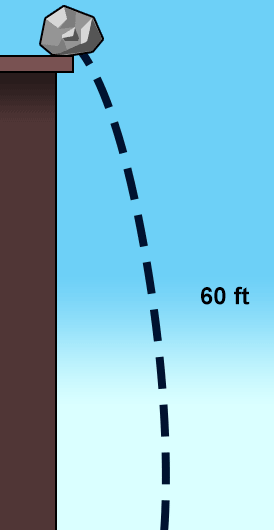
The rock in the picture is pushed over the edge and it begins to fall. Which type of energy conversion is taking place?
mechanical to heat
sound to mechanical
potential to kinetic
kinetic to potential
9
Drag and Drop

10

State of Matter: Solids, liquids, and gases transfer energy differently.
Amount of Matter: More mass = more energy needed to change temperature.
Environment: Heat transfer depends on surroundings.
Factors Affecting Heat Transfer
11
Multiple Choice
Which factor affects the amount of energy needed to change the temperature of a substance?
The color of the substance
The shape of the container
The mass of the substance
The presence of oxygen
12
Multiple Choice
Why does a large pot of water take longer to heat than a small one?
It absorbs heat faster
It reflects heat away
It has more mass, requiring more energy transfer
It contains less matter
13
Measuring Energy Transfer
Temperature is a measure of the average kinetic energy of particles.
Heat moves from warmer objects to cooler objects until equilibrium is reached.
Example: Ice melting in warm water.
14
Multiple Choice

15
Multiple Choice
What are the units of energy?
Watt
Newton
Joule
Kelvin
16
Multiple Choice
Energy that is stored in a system, ready to be used, is called ____________ energy.
Kinetic
Potential
Conserved
Power
17
Multiple Choice
Energy in a system that is moving is called _______________ energy.
Kinetic
Potential
Mechanical
Power
18
Multiple Choice

During the summer, Chang takes a glass of milk from the refrigerator and places it on the kitchen counter. Over time, energy as heat is transferred to the milk from the air. Which change would most likely occur?
The temperature of the milk will increase.
The temperature of the milk will decrease.
The milk will change from a liquid to a gas.
The milk will change from a liquid to a solid.
19
Multiple Choice
How does temperature relate to kinetic energy?
Higher temperature means lower kinetic energy
Higher temperature means higher kinetic energy
Temperature and kinetic energy are unrelated
Kinetic energy remains constant regardless of temperature
20
Multiple Choice
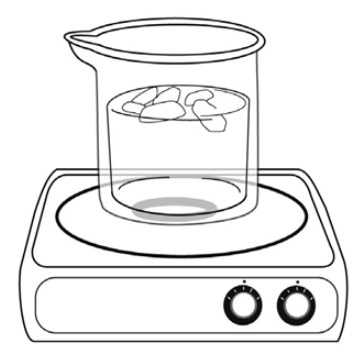
Emerson places a beaker of water on a hotplate and turns the hotplate on. The temperature in the room is 25 ºC. After the water heats to 75 ºC, she adds pieces of ice to the beaker.
Which of the following describes a transfer of heat energy that occurs during this experiment?
from the air to the water
from the ice to the beaker
from the water to the ice
from the water to the hotplate
21
Chemical Energy
potential energy
depends on the kinds of atoms and their arrangement
batteries
matches
breakdown of food


22
Electrical Energy
energy results from position or motion of charged particles
how we get light from power lines

23
Sound Energy
kinetic energy
caused by vibration of particles in a medium such as steel, water, or air

24
Thermal Energy
kinetic energy
particles move faster the higher the temperature

25
Electromagnetic Energy
transmitted through space in the form of electromagnetic waves
visible light
x-rays
microwaves

26
Real World Examples
Cooking food: Heat transfers from stove to pan to food.
Solar panels: Convert light energy to electrical energy.
Rubbing hands together: Converts kinetic energy to heat.
Transfer of Energy
8.SC.15 Use models to construct an explanation of how energy is transformed but still conserved.
By NaaJah Richardson
Show answer
Auto Play
Slide 1 / 26
SLIDE
Similar Resources on Wayground

20 questions
Earth's Changing Climate
Lesson
•
6th Grade

20 questions
Fossil Fuels
Lesson
•
6th Grade

19 questions
Energy Transfer and the Water Cycle
Lesson
•
6th Grade

22 questions
Physical and Chemical Weathering
Lesson
•
6th Grade

22 questions
Taxonomy - Domains and Kingdoms Notes
Lesson
•
6th Grade

18 questions
Variables Practice Problems
Lesson
•
6th Grade

20 questions
Force, Work and Energy
Lesson
•
5th - 6th Grade

20 questions
Nervous & Circulatory System
Lesson
•
5th - 6th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

13 questions
SMS Cafeteria Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

12 questions
SMS Restroom Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

10 questions
Pi Day Trivia!
Quiz
•
6th - 9th Grade
Discover more resources for Science

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
Cell Organelles and Functions
Quiz
•
6th - 8th Grade

10 questions
Exploring the Layers of the Earth
Interactive video
•
6th - 10th Grade

21 questions
Waves and Wave Properties
Quiz
•
6th - 8th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

13 questions
Plate Tectonics and Earth's Structure
Lesson
•
6th - 8th Grade

10 questions
Exploring Heat Transfer: Conduction, Convection, and Radiation
Interactive video
•
6th - 8th Grade

20 questions
Amplify Weather Patterns Chapter 1
Quiz
•
6th Grade