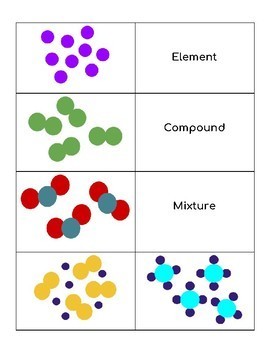
Atoms Elements Compounds Mixtures
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Standards-aligned
James Gonzalez
FREE Resource
14 Slides • 7 Questions
1
2
Multiple Select
3
4
5
6
7
8
9
Multiple Select
10
Multiple Choice
11
12
Multiple Choice
13
14
Multiple Select
15
16
17
18
Multiple Choice
19
Multiple Choice
20
21
Show answer
Auto Play
Slide 1 / 21
SLIDE
Similar Resources on Wayground

17 questions
Rate of Reaction & Collision Theory
Lesson
•
9th - 12th Grade

19 questions
Chemical Bonding
Lesson
•
9th - 12th Grade

15 questions
Significant Figures
Lesson
•
10th - 11th Grade

17 questions
Subscripts and Coefficients
Lesson
•
9th - 12th Grade

16 questions
Chemistry Unit 7 - Stoichiometry Day 1 Notes
Lesson
•
9th - 12th Grade

16 questions
Stem Changing Verbs
Lesson
•
9th - 12th Grade

16 questions
Affirmative & Negative Words
Lesson
•
9th - 12th Grade

16 questions
Lesson: Solving One-Step Equations
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

10 questions
Probability Practice
Quiz
•
4th Grade

15 questions
Probability on Number LIne
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

6 questions
Appropriate Chromebook Usage
Lesson
•
7th Grade

10 questions
Greek Bases tele and phon
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Predicting Products
Quiz
•
9th - 12th Grade

11 questions
Balancing Chemical Equations
Lesson
•
9th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

19 questions
Stoichiometry, % yield, Limiting Reactants
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

10 questions
Exploring Ionic and Covalent Bonding Concepts
Interactive video
•
6th - 10th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

24 questions
Unit 2 (Part 1) Bonding Review
Quiz
•
12th Grade