

Molecular Structures
Presentation
•
Chemistry
•
10th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
10 Slides • 4 Questions
1
Giant Molecular Structures
Cambridge AS Level Chemistry

2
Giant Molecular Structures
Some covalently bonded structures have a three dimensional network of covalent bonds that called giant molecular structures or giant covalent structures.
They have high melting and boiling points because of the large number of strong covalent bonds linking the whole structure.
3
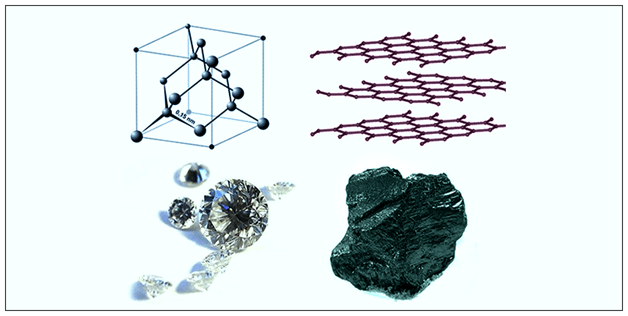
Giant molecular structures can be elements (e.g. graphite and diamond), and can be a compound, such as silicon oxide.
Diamond and graphite are different forms of the same element. Different crystalline or molecular forms of the same element are called allotropes.
4
Multiple Choice

Define the term allotropes
Atoms with same proton number but different number of neutrons
Atoms with same proton number but different mass number
Atoms in different arrangement in structure
Atoms in different arrangement and different physical form
5
Graphite
In graphite, the carbon atoms are arranged in planar layers. Within the layers, the carbon atoms are arranged in hexagons. Each carbon atom is joined to three other carbon atoms by strong covalent bonds.
The fourth electron of each carbon atom occupies a p orbital. These p orbitals overlap sideways. A cloud of delocalised electrons is formed above and below the plane of the carbon rings.
The layers of carbon atoms are kept next to each other by weak van der Waals forces.

6
Physical properties of graphite
High melting and boiling points: there is strong covalent bonding throughout the layers of carbon atoms. A lot of energy is needed to overcome these strong bonds.
Softness: graphite is easily scratched. The forces between the layers are weak. The layers of graphite can slide over each other when force is applied. The layers readily flakes off. This is why graphite is used in 'pencil leads' and feels slippery.
Good conductor and electricity: when a voltage is applied, the delocalised electrons (mobile electrons) can move along the layers.
7
Open Ended
Why graphite can conduct electricity?
8
Diamond
In diamond, each carbon atom forms four covalent bonds with other carbon atoms. The carbon atoms are tetrahedrally arranged around each other. The network of carbon atoms extends almost unbroken throughout the whole structure. The regular arrangement of the atoms gives diamond a crystalline structure.

9
Physical properties of diamond
High melting and boiling points: there is strong covalent bonding throughout the whole structure. A lot of energy is needed to break these bonds and separate the atoms.
Hardness: diamond cannot be scratched easily because it is difficult to break the three-dimensional network of strong covalent bonds.
Does not conduct electricity or heat: each of the four outer electrons on every carbon atom is involved in covalent bonding. This means that there are no free electrons available to carry the electric current.
10
Multiple Choice
Which answer shows the correct number of C-C covalent bonds in its allotropes?
Diamond 3, Graphite 3
Diamond 4, Graphite 3
Diamond 3, Graphite 4
Diamond 4, Graphite 4
11
Silicon (IV) oxide, SiO2
There are several forms of silicon (IV) oxide. The silicon (IV) oxide found in mineral quartz has a structure similar to diamond.

12
The structure of SiO2
Each silicon atom is bonded to four oxygen atoms but each oxygen atom is bonded to only two silicon atoms. So the formula for silicon (IV) oxide is SiO2.

13
Physical properties of silicon (IV) oxide
Silicon dioxide has properties similar to that of diamond. It forms hard, colourless crystals with high melting and boiling points and it does not conduct electricity.
Sand is largely silicon (IV) oxide.
14
Multiple Choice
Silicon (IV) oxide has properties that similar to graphite.
TRUE
FALSE
Giant Molecular Structures
Cambridge AS Level Chemistry

Show answer
Auto Play
Slide 1 / 14
SLIDE
Similar Resources on Wayground

13 questions
Chapter 6 Section 5 Part 1: Molecular Geometry VSEPR
Presentation
•
10th - 12th Grade

12 questions
Full stoich practice
Presentation
•
10th - 12th Grade

12 questions
NGSS MS PS1-2
Presentation
•
10th - 12th Grade

12 questions
Molecular Empirical Lesson
Presentation
•
10th - 12th Grade

13 questions
Remediation day 1
Presentation
•
10th - 12th Grade

11 questions
IMFS Lesson
Presentation
•
9th - 12th Grade

13 questions
Density and States of Matter Lesson
Presentation
•
10th - 12th Grade

14 questions
L1 Types of Matter
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Chemistry

15 questions
Balancing and Reaction Types
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

16 questions
STAAR Review Quizziz 5
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

5 questions
DOL REC: pH and pOH Calculations
Quiz
•
10th - 11th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

5 questions
DOL PreAP: pH and pOH Calculations
Quiz
•
10th Grade

13 questions
IP PREAP: pH and pOH Calculations
Quiz
•
10th - 11th Grade