

Mixtures and Compounds
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Standards-aligned
Joseph Anderson
FREE Resource
14 Slides • 7 Questions
1
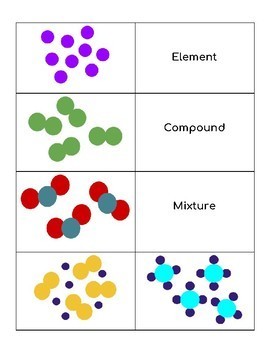
Elements, Compounds, Mixtures
Definitions , Examples, Comparisons
2
Multiple Select
(check all that apply)
You...
have mass.
have volume.
matter.
3
Chemistry is the study of matter
Matter is anything that has mass and occupies space (has volume)
Composition
physical properties
chemical properties

4
All matter is composed of atoms.
Atoms are the "building blocks" of the universe

5
Matter exists in 4 fundamental states (a.k.a. 4 phases)
solid
liquid
gas
plasma

6
All matter is either a pure substance or a mixture of substances.
(Most everyday items are mixtures)

7
A pure substance has uniform composition and properties.
Every portion of a pure substance looks and acts the same.
Can be an element or a compound.

8
An Element
A substance composed of only one type of atom.
Multiple atoms of the same type can be bonded together.
Elements cannot be broken down by chemical means.

9
Multiple Select
Which of the following are elements?
He
O2
S8
Fe2O3
O3
10
Multiple Choice
Hydrogen gas, H2, can be broken down by chemical means.
True
False
11
A Compound
A substance composed of 2 or more elements chemically bonded.
Component elements have a definite ratio (E.g. 2:1 for H:O in water)
Compounds can be separated by chemical means into their component elements.

12
Multiple Choice
Water, H2O, can be broken down by chemical means.
True
False
13
Life
Mixing it up since a while ago

14
Multiple Select
Which of the following are mixtures
tap water
bread flour
blood
beach sand
15
A Mixture
A physical combination of 2 or more pure substances

16
Mixtures
Component substances can be separated by physical means.
Component substances retain their original properties.

17

18
Multiple Choice

Which of the following is a mixture?
(circles of different colors represent different types of atoms)
(circles that are touching are chemically bonded)
19
Multiple Choice

Which of the following is a compound?
(circles of different colors represent different types of atoms)
(circles that are touching are chemically bonded)
20

21
Recap
Matter is anything with mass and volume.
Matter is made of atoms.
Matter can be pure or a mixture.
Elements are made of only one type of atom. Cannot be chemically broken down.
Compounds are made of 2 or more types of atoms. Can be chemically broken down.

Elements, Compounds, Mixtures
Definitions , Examples, Comparisons

Show answer
Auto Play
Slide 1 / 21
SLIDE
Similar Resources on Wayground

15 questions
Reading solubility curves
Presentation
•
9th - 12th Grade

19 questions
Ionic Compounds
Presentation
•
9th - 12th Grade

13 questions
Classification of Matter
Presentation
•
9th - 12th Grade

14 questions
Particle Diagrams
Presentation
•
9th - 12th Grade

20 questions
Properties of Matter Notes
Presentation
•
9th - 12th Grade

12 questions
Experience 3: Atomic Emission Spectra
Presentation
•
9th - 12th Grade

14 questions
Periodic Table and Trends
Presentation
•
10th - 12th Grade

15 questions
Nuclear Fission vs Nuclear Fusion
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Chemistry

15 questions
Balancing and Reaction Types
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

16 questions
STAAR Review Quizziz 5
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

5 questions
DOL REC: pH and pOH Calculations
Quiz
•
10th - 11th Grade

11 questions
Balancing Chemical Equations
Presentation
•
9th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

5 questions
DOL PreAP: pH and pOH Calculations
Quiz
•
10th Grade