

Periodic Table
Presentation
•
Science
•
6th - 8th Grade
•
Medium
Joseph Anderson
Used 1+ times
FREE Resource
23 Slides • 23 Questions
1
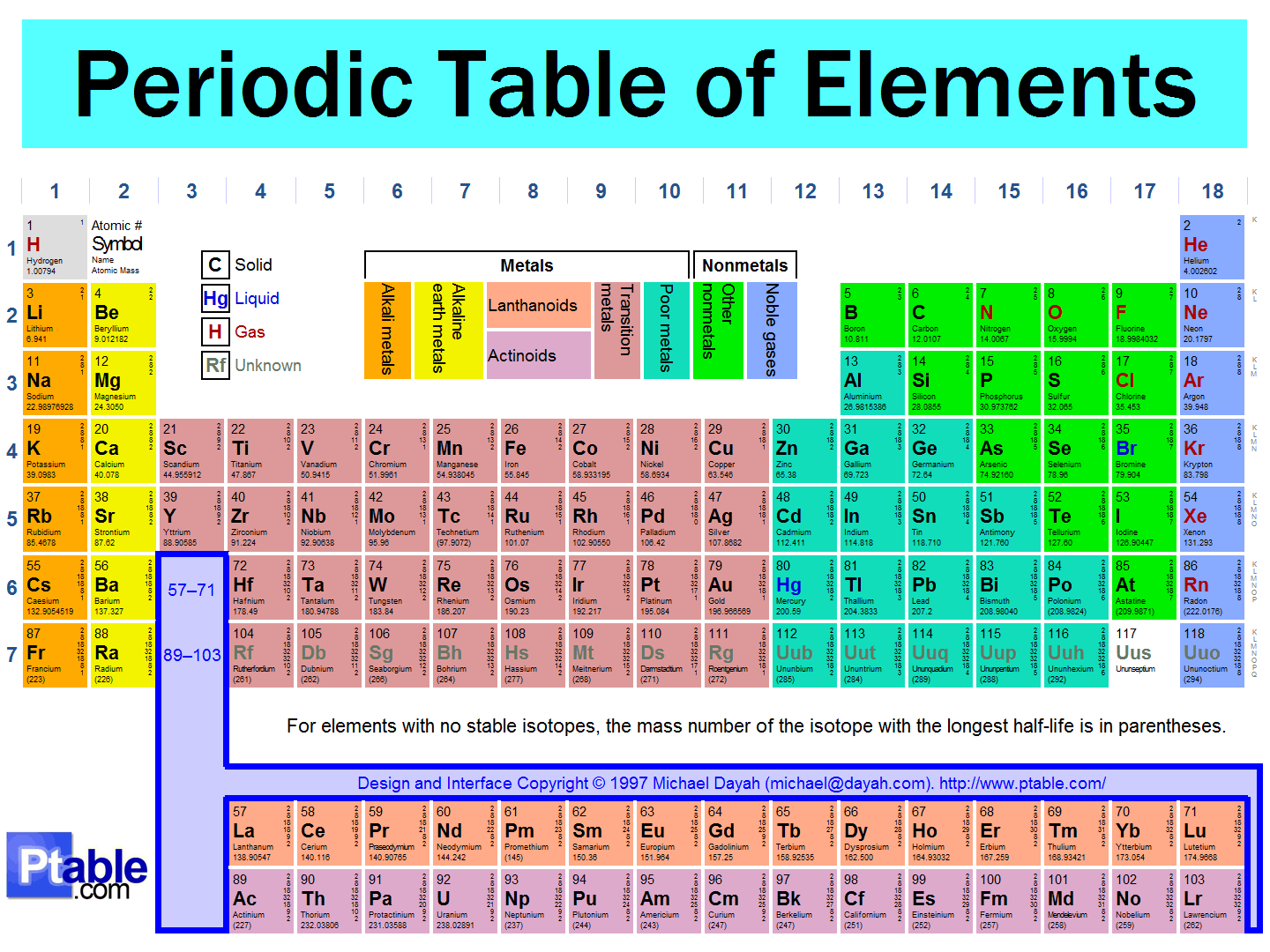
The Periodic Table

2
Classifying Matter
Elements: pure substances, on the Periodic Table
Compounds: two or more elements; parts on the Periodic Table
Mixtures: two or more substances MIXED; not on Periodic Table
3

4
Periodic Table
8.P.1.2 Explain how the physical properties of elements and their reactivity have been used to produce the current model of the Periodic Table of elements.

5

6

7
Multiple Select
Choose all the items that are elements.
NaCl (table salt)
Sulfur
O2
Water
Air
8
Multiple Select
Choose all of the items that are compounds.
NaCl (table salt)
Sulfur
O2
Water
Air
9
Multiple Select
Choose all of the items that are mixtures
NaCl (table salt)
Sulfur
O2
water
Air
10
Groups & Periods
Groups: Columns; up & down, similar properties. ⬆️ ⬇️
Periods: Rows; left to right; increasing atomic ➡️ ⬅️

11

12

13

14
Multiple Choice
Which element is similar to Li?
Be
K
15
Multiple Choice
Which element is similar to Mg?
Ca
B
16
Multiple Choice
Which element is similar to B?
C
Al
17
Multiple Choice
Which element is similar to F?
Ne
Cl
18
Multiple Choice
Which element is similar to He?
H
Ar
19
Reactivity
ability of a substance to react
decreases left ➡️ to right ⬅️
Group 1 Alkali Metals are most reactive
Group 18 Noble Gases are least reactive, inert

20
Multiple Choice
Which element is the least reactive?
Li
Ne
B
C
21
Multiple Choice
Which element is the least reactive?
Mg
Na
C
S
22
Multiple Choice
Which choice shows the elements in order from most reactive to least reactive. Sr, K, Cl, P
Sr, K, Cl, P
K, Cl, Sr, P
K,P, Sr, Cl
K, Sr, P, Cl
23
Multiple Choice
Which best describes the sequence of the elements in Period 5 of the periodic table?
highly reactive gases, relatively nonreactive metals, metalloids, very nonreactive gases, nonreactive nonmetals
highly reactive metals, relatively nonreactive metals, metalloids, reactive nonmetals, nonreactive gases
nonreactive gases, metalloids, nonreactive metals, highly reactive metals, highly reactive gases
nonreactive gases, nonreactive metals, highly reactive metals, metalloids,
reactive nonmetals, reactive gases
24
Multiple Choice
Which group of the periodic table is composed of inert (not reactive) gases?
answer choices
alkali metals
alkaline earth metals
halogens
noble gases
25

26

27
Periodic Table Periods:
Periods run in rows.
Each period has the same number of electron shells. i.e. Hydrogen and Helium have 1 electron shell. Lithium and Boron have 2.
This means each period has the same number of ENERGY LEVELS!

28

29

30
Multiple Choice
31
Multiple Choice

32
Multiple Choice
Groups run:
up and down
left to right
33
Multiple Choice

Which two elements share common characteristics based on the periodic table?
Sodium and Magnesium
Chlorine and Sulfur
Argon and Neon
Calcium and Bromine
34
Multiple Choice

How are the 3 elements in the picture organized on the periodic table?
Niobium and cobalt have the same energy levels.
Cobalt and scandium have the same energy levels.
Cobalt and scandium have the same characteristics.
Niobium and cobalt have the same characteristics.
35
Multiple Choice

How are the 3 elements in the picture organized on the periodic table?
Niobium and cobalt have the same energy levels.
Cobalt and scandium have the same energy levels.
Cobalt and scandium have the same characteristics.
Niobium and cobalt have the same characteristics.
36
Metals, Nonmetals, Metalloids
Metals: ductile, malleable, luster, solids
Metalloids: properties of metals & nonmetals
Nonmetals: brittle & dull

37
Metals, Metalloids, and Nonmetals
The periodic table is divided into 3 types of chemicals
They are in alphabetical order: 1. Metal, 2. Metalloid, 3. Nonmetal
Metals are the largest group
Metalloids are the smallest group between metals and nonmetals.

38
Metals
Metals are on the left side of the table.
Characteristics include: malleable, ductile, shiny, and conductive (electricity and heat)

39
Metalloids
Metalloids are on the stairstep between metals and nonmetals.
Characteristics include: somewhat malleable, a little ductile, somewhat shiny, and conductive at high temperatures

40
Nonmetals
Nonmetals are on the right side of the table.
Dull, brittle, non-conductors

41
Multiple Choice
Students in Mrs. Clark's science class are investigating a substance that has the following properties
-shiny
-bends easily
-hard, not easily broken
Based on these properties how would you classify the substance?
metal
nonmetal
metalloid
42
Multiple Choice
Metals are on the:
Left side
Right Side
Stairstep
43
Multiple Choice
Metalloids are on the:
Left side
Right Side
Stairstep
44
Multiple Choice
Nonmetals are on the:
Left side
Right Side
Stairstep
45

46
Song
The Periodic Table

Show answer
Auto Play
Slide 1 / 46
SLIDE
Similar Resources on Wayground

43 questions
Craig Describing Matter
Presentation
•
6th - 8th Grade

44 questions
3.2 Metals and Oxygen
Presentation
•
7th Grade

35 questions
Properties of Matter Review
Presentation
•
6th - 9th Grade

47 questions
M1-บทที่ 4 วัสดุและเครื่องมือช่าง
Presentation
•
7th Grade

47 questions
Structures of Life
Presentation
•
6th - 8th Grade

41 questions
Atoms and Elements
Presentation
•
6th - 8th Grade

35 questions
Magnetic Force
Presentation
•
6th - 8th Grade

41 questions
Metals Nonmetals Metalloids
Presentation
•
7th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Abiotic & Biotic Factors
Quiz
•
7th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade