

Chemistry Test Review
Presentation
•
Science
•
7th - 9th Grade
•
Hard
Joseph Anderson
FREE Resource
5 Slides • 40 Questions
1
Chemical reaction test review

2
Atomic structure
3
Multiple Choice
Subatomic particles with a negative charge
Electrons
Neutrons
Protons
Quarks
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice

The smallest particle of an element.
Element
Molecule
Atom
9
Multiple Choice

What is the atomic mass of Neon?
10
20
30
10
Multiple Choice

What is one way Neutrons are different from electrons?
they both are negative
neutrons have no charge, while electrons have a negative charge
Both are in the nucleus( center of atom)
Electrons are inside nucleus and neutrons are outside the nucleus.
11
Multiple Choice

What is the atomic number?
35
18
17
52
12
Multiple Choice

How many protons and electrons does this element have?
35
18
17
52
13
Multiple Choice

How many neutrons does Chlorine have?
18
17
35
52
14
Multiple Choice

What is the atomic mass of Arsenic?
33
107
75
15
Multiple Choice

How many neutrons does calcium have?
20
40
60
0
16
Couting Atoms
17
Multiple Choice
How many atoms of Hydrogen are in H2O
1
2
3
more than 3
18
Multiple Choice
How many different elements are present in H2COCH2
5
4
3
2
19
Multiple Choice
Calculate the number of Oxygen atoms in Al(OH)3
1
2
3
4
20
Multiple Choice
The ______ are numbers found on the lower right-hand side of an element symbol.
Coefficient
Exponent
Multiplier
Subscript
21
Multiple Choice
Coefficients in chemical formulas...
Multiply just the first atom
Add to the subscripts at the end of the molecule
Multiply elements without subscripts
Multiply EVERYTHING!
22
Multiple Choice
4CF2Cl2
C = 4, F = 4, Cl = 2
C = 1, F = 8, Cl = 8
C = 4, F = 8, Cl = 8
C = 1, F = 4, Cl = 8
23
Multiple Choice
C3H5(NO3)3
C = 3, H = 5, N = 3, O = 9
C = 3, H = 5, N = 1, O = 9
C = 9, H = 15, N = 3, O = 9
C = 9, H = 15, N = 1, O = 3
24
Multiple Choice
H2SO4
H = 2, S = 4, O = 4
H = 2, S = 1, O = 4
H = 8, S = 4, O = 4
H = 2, SO = 4
25
Multiple Choice
How many different elements are present in this molecule?
3 NH4C2H3O2
15
36
5
4
26
Multiple Choice
A ______ is a number written in front of a chemical formula.
Coefficient
Subscript
Exponent
Coordinator
27
The law of conservation of mass
28
Multiple Choice
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice
32
Multiple Choice
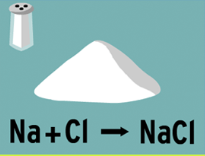
When sodium and chlorine combine to form sodium chloride, sodium chloride is the:
Originator
Reactant
Product
Produce
33
Multiple Choice

34
Multiple Choice
Law of conservation of mass states the amounts of atoms in the reactants and the products...
Increase
Decrease
Stay the same
No correct answer
35
Multiple Choice
Does the equation H2+O2 →H2O follow the Law of Conservation of Mass?
Yes, because the atoms are equal.
No, because more info is needed.
No, because there are more atoms in the product.
No, because there are more atoms in the reactant.
36
Multiple Choice
37
Endothermic and Exo thermic
38
Multiple Choice

39
Multiple Choice

40
Multiple Choice

41
Multiple Choice

42
Multiple Choice
02 (g)+ 2H2-------------2H20 + Energy
endothermic
exothermic
43
Multiple Choice
6CO2 + 6H20 + Energy --------------- C6 H12 06 + 6O2
endothermic reaction
exothermic reaction
44
Multiple Choice

_____ reactions usually feel hot!
endothermic
exothermic
45
Multiple Choice

____ reactions usually feel cold.
endothermic
exothermic
Chemical reaction test review

Show answer
Auto Play
Slide 1 / 45
SLIDE
Similar Resources on Wayground

39 questions
Weathering and Erosion
Presentation
•
6th - 8th Grade

38 questions
3.5 Mixtures and Solutions
Presentation
•
8th Grade

38 questions
Hydrosphere Review
Presentation
•
8th Grade

43 questions
Into to waves
Presentation
•
7th - 9th Grade

37 questions
Thermal Energy
Presentation
•
6th - 8th Grade

37 questions
Physics STAAR Review - Graphing & Calculations
Presentation
•
6th - 8th Grade

38 questions
Forms of Energy
Presentation
•
7th - 8th Grade

39 questions
MENDELIAN GENETICS (PART 3)
Presentation
•
7th - 10th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Science

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Abiotic & Biotic Factors
Quiz
•
7th Grade

18 questions
Conduction Convection Radiation
Quiz
•
7th Grade