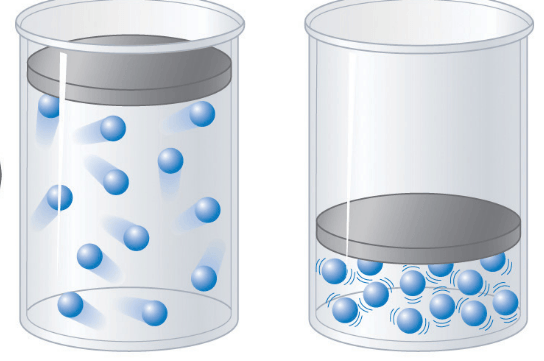
Dalton's Partial Pressure
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
7 Slides • 10 Questions
1
2
3
4
5
6
7
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

12 questions
Word Choice,: Figurative, Connotative, and Impact
Lesson
•
9th - 12th Grade

11 questions
Teenager 5 Session 22: Pronunciation & Speaking
Lesson
•
KG

11 questions
Inverse Trig Functions
Lesson
•
9th - 12th Grade

11 questions
Map Skills In History
Lesson
•
9th - 12th Grade

13 questions
Congruent Triangles
Lesson
•
9th - 12th Grade

13 questions
Spanish Double Object Pronouns
Lesson
•
9th - 12th Grade

12 questions
Significant Digits
Lesson
•
9th - 12th Grade

13 questions
Ch. 5.3 - Physics and the Quantum Mechanical Model
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

10 questions
Probability Practice
Quiz
•
4th Grade

15 questions
Probability on Number LIne
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

6 questions
Appropriate Chromebook Usage
Lesson
•
7th Grade

10 questions
Greek Bases tele and phon
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Predicting Products
Quiz
•
9th - 12th Grade

11 questions
Balancing Chemical Equations
Lesson
•
9th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

19 questions
Stoichiometry, % yield, Limiting Reactants
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

10 questions
Exploring Ionic and Covalent Bonding Concepts
Interactive video
•
6th - 10th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

24 questions
Unit 2 (Part 1) Bonding Review
Quiz
•
12th Grade