

Holt Ch 1 Guided Notes
Presentation
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Easy
+4
Standards-aligned
Connie Schaef
Used 6+ times
FREE Resource
72 Slides • 74 Questions
1

Chapter 1
2

Chapter 1
3

Chapter 1
4

Chapter 1
5

Chapter 1
6

Chapter 1
7

Chapter 1
8
In an experiment, there are two main variables: The independent variable: the variable that an experimenter changes or controls so that they can observe the effects on the dependent variable. The dependent variable: the variable being measured in an experiment that is “dependent” on the independent variable.
For example, a researcher might change the amount of water they provide to a certain plant to observe how it affects the growth rate of the plant.
In this example, the amount of water given to the plant is controlled by the researcher and, thus, is the independent variable. The growth rate is the dependent variable because it is directly dependent on the amount of water that the plant receives and it’s the variable we’re interested in measuring.
9
Multiple Choice
Information obtained through the senses.
Observation
Hypothesis
Principle
Inference
10
Multiple Choice
The condition in an experiment that is changed or manipulated.
Dependent Variable
Experimental Group
Independent Variable
Control Group
11
Multiple Choice
The condition in an experiment that is observed or measured
Dependent Variable
Experimental Group
Independent Variable
Control Group
12
Multiple Choice
In an experiment the group that serves as a standard for comparison and receives no experimental treatment.
Dependent Variable
Experimental Group
Independent Variable
Control Group
13
Multiple Choice
In an experiment the group that receives the experimental treatment.
Dependent Variable
Experimental Group
Independent Variable
Control Group
14
Multiple Choice
The condition that does not change in an experiment.
Data
Inference
Variable
Constant
15
Multiple Choice
Data expressed in words.
Qualitative Data
Quantitive Data
16
Multiple Choice
Data expressed in numbers.
Qualitative Data
Quantitive Data
17
Multiple Choice
Identify the independent variable in the following hypothesis.
If more salt is added to water then the water will take longer to boil because salt causes the water to need more heat to boil.
More heat is needed.
Amount of salt
Time to boil
None of these
18
Multiple Choice
Identify the dependent variable in the following hypothesis.
If more salt is added to water then the water will take longer to boil because salt causes the water to need more heat to boil.
More heat is needed.
Amount of salt
Time to boil
None of these
19
Multiple Select
Which of the following are considered steps in the scientific method?
Observing
Formulating Hypotheses
Testing
All of the above
20
Mystery Solution Match Up
Objective: Students will be given 3 colorless mystery solutions with the same solutions as their lab partner has? There is one catch -- once a course of action has been determined, no visual communication is allowed!
Lab Partner: Matching Animals
21

Chapter 1
22

23

Chapter 1
24

Chapter 1
25

Chapter 1
26

Chapter 1
27

Chapter 1
28

Chapter 1
29
Metric Prefixes
and conversions
30
King Henry Died By drinking chocolate milk
King Henry Died By drinking chocolate milk
31
Multiple Choice
32
Multiple Choice
Convert 17 m to dm:
1.7 dm
0.017 dm
1,700 dm
170 dm
33
Multiple Choice
34
Multiple Choice
35
Multiple Choice
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
Which SI unit would you use to measure the length of a football field?
Meter
Kilogram
Second
Candela
44

Chapter 1
45

Chapter 1
46

Chapter 1
47

Chapter 1
48

Chapter 1
49

Chapter 1
50

Chapter 1
51

Chapter 1
52

Chapter 1
53
Multiple Select
Which of the following are examples of conversion factors as shown in the slides?
4 quarters/1 dollar
1 dollar/4 quarters
0.25 dollar/1 quarter
1 quarter/0.25 dollar
54

Chapter 1
55

Chapter 1
56

Chapter 1
57
Fill in the Blanks
58

Chapter 1
59

Chapter 1
60

Chapter 1
61
You are given a block of metal that has a ‘known’ mass of 110.2 grams. You measure its mass three times and get the following mass readings: 100.2 g, 100.1 g 100.3g. Describe in terms of accurate and precise.
62
Multiple Choice

Student A
Student B
Student C
Cannot be determined
63
Multiple Choice

64
Multiple Choice

This bullseye demonstrates...
High Accuracy & High Precision
High Accuracy & Low Precision
Low Accuracy & High Precision
Low Accuracy & Low Precision
65
Multiple Choice

Describe the accuracy and precision of the image
Very accurate and somewhat precise
Very accurate but not precise at all
Not accurate at all but very precise
not accurate and not very precise
66
Multiple Select
A set of data are all close in value to each other, but they are not close to the actual value. This set of data can be described as _________________.
precise
accurate
67
Multiple Choice
Accuracy means
the measurements are close to each other
the measurement is close to the true value
the measurements are close to each other
the measurements are close to each other and the true value.
68
Multiple Choice
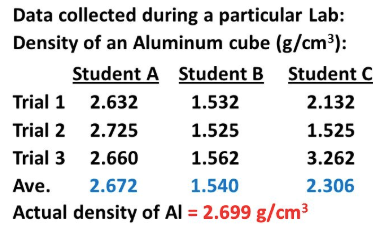
Which student is the most ACCURATE?
A
B
C
69
Multiple Choice

True
Choose the other answer
70
Multiple Choice
A company claims to sell 15 candy pieces in each bag. Judy counts her bag and finds 14, Josh finds 16, and Jill finds 15. The company is...
accurate
precise
accurate and precise
neither accurate nor precise
71
Multiple Choice
Measurements: 2.5, 14.5, 10.1, 45.3
True Value: 25.0
These measurements are…
Accurate, but not precise
Precise, but not accurate
Both precise and accurate
Neither precise nor accurate
72

Chapter 1
73
You are given a block of metal that has a ‘known’ mass of 110.2 grams. You measure its mass three times and get the following mass readings: 100.2 g, 100.1 g 100.3g. What is your percent error?
74

Chapter 1
75

Chapter 1
76
An error in chemistry still often means a mistake turn and talk to your neighbor about the type of things that might lead to an error.
77
Reading a scale incorrectly
Misreading gauges
Making math mistakes during dilutions and other types of calculations
Spilling chemicals during transfer
Incorrect or non-existent calibration of instruments
Limitations of measurement equipment in a lab as a source of error.
78

Chapter 1
79

Chapter 1
80
Rule 1 - Non-zero digits are ALWAYS significant This is a fundamental rule and the easiest to understand. Any non-zero digit in a number must be considered as significant.
Examples:
1.1 has two significant figures (1, 1). 13.55 has four significant figures (1, 3, 5, 5).
81
Rule 2 - any zero contained between two non-zero numbers is significant
Examples:
1.05 has three significant figures (1, 0, 5); the zero is enclosed by non-zero digits and should be counted.
100.45001 has eight significant figures (1, 0, 0, 4, 5, 0, 0, 1); all the zeroes in this number are enclosed by non-zero digits and therefore should be counted.
82
Rule 3 - leading zeroes are never significant Any leading zeroes are never significant, irrespective of a decimal point
Examples:
0.05 - one significant figure (5); the leading zeroes are ignored.
0.0501 - three significant figures (5, 0, 1); the leading zeroes are ignored, the third zero is enclosed by two non-zero digits and is therefore significant.
83
Rule 4 - final or trailing zeroes are significant only after a decimal point Zeroes contained before or after a decimal point are considered as significant figures if they fall between two non-zero digits, as per rule 2, OR potentially when they are trailing, not leading zeroes. Trailing zeroes are only counted when a decimal point is included in the number, as shown below.
84
1.005 - four significant figures (1, 0, 0, 5); the zeroes are automatically counted as they fall between two non-zero digits. 0.005 - one significant figure (5); the leading zeroes do not fall between two non-zero digits and only the 5 is considered significant.
0.005 - one significant figure (5); the leading zeroes do not fall between two non-zero digits and only the 5 is considered significant.
85
0.00500 - three significant figures (5, 0, 0); the leading zeroes do not count (as per Rule 3) but the two trailing zeroes are considered to be significant as they come after a decimal point. So in this example the three significant figures are the 5 and the final two zeroes.
86
500 - one significant figure (5); there is no decimal point and therefore the two zeroes do not add anything to the "precision" of the number. However, if a decimal point is present then these zeroes will become significant, as per example 500.
500.00 - five significant figures (5, 0, 0, 0, 0). The four trailing zeroes are all significant due to the decimal point which adds precision to the number.
87

Chapter 1
88

Chapter 1
89

Chapter 1
90

Chapter 1
counting numbers are exact so you would say infinite because counting numbers go to infinity
91
Multiple Choice
92
Multiple Choice
Significant digits contain all of the digits that are certain plus _____________ estimated digit(s).
0
1
2
3
93
Multiple Choice
Significant digits are used in chemistry to show the _____________ of the measuring tool.
units
purpose
precision
94
Multiple Choice
95
Multiple Choice
96
Multiple Choice
97
Multiple Choice
98
Multiple Choice
99
Multiple Choice
100
Multiple Choice
101
Multiple Choice
102
Multiple Choice
103
Multiple Choice
104

Chapter 1
105

Chapter 1
106

Chapter 1
107
Multiple Choice
Subtract and round to the appropriate number of significant digits. 65.76−11.3
54.46
54.4
54.5
54
108
Multiple Choice
23.91 x 12.861 = 307.50651
109
Multiple Choice
65 ÷ 10.01= 6.4935064
110
Multiple Choice
What is the answer expressed with the correct significant digits for: 6.00 cm + 3.411 cm?
9.41 cm
9.411 cm
9.4 cm
9 cm
111
Multiple Choice
What is the answer expressed with the correct significant digits for: 3.4 cm x 2.32 cm?
7.888 cm2
7.89 cm2
7.9 cm2
8 cm2
112
Multiple Choice
What is the answer expressed with the correct significant digits for:
7.77 g÷2.3 cm3
3.37826 g/cm3
3.38 g/cm3
3.4 g/cm3
3.378 g/cm3
113

Chapter 1
114

Chapter 1
115
Multiple Choice
116
Multiple Choice
117
Multiple Choice
118
Multiple Choice
119
Multiple Choice
540,020,000
120
Multiple Choice
0.0000378
121
Multiple Choice
0.0079245
122
Multiple Choice
123
Multiple Choice
124

Chapter 1
125

Chapter 1
126

Chapter 1
127
Multiple Choice
(6.9 x 107) / (2.3 x 10-3)
128
Multiple Choice
(9.4 x 106)(3.2 x 105)
129
Multiple Choice

130

Chapter 1
131

Chapter 1
132

Chapter 1
133

Chapter 1
134

Chapter 1
135

Chapter 1
136

Chapter 1
137
Multiple Choice
138
Multiple Choice
139
Multiple Choice
140
Multiple Choice
141
Multiple Choice
142
Multiple Choice
A crayon has a mass of 10.0 g and volume of 5.0 cm3. What is the density?
30 g/cm3
1.0 g/cm3
15 g/cm3
2.0 g/cm3
143
Multiple Choice
Find the mass of 250 mL of benzene that has a density of .8765 g/mL.
220 mL
290 g
220 g
219.13 g
144

Chapter 1
145

Chapter 1
146

Chapter 1

Chapter 1
Show answer
Auto Play
Slide 1 / 146
SLIDE
Similar Resources on Wayground

141 questions
ΧΗΛΕΙΑ Α
Presentation
•
9th Grade

141 questions
Survival English
Presentation
•
KG - University

137 questions
World History: Chapter #3 Powerpoint Lesson
Presentation
•
9th - 12th Grade

135 questions
Mes lecons
Presentation
•
KG - University

144 questions
MODUL GENERATIVE AI
Presentation
•
10th Grade

136 questions
Cold War
Presentation
•
11th Grade

138 questions
WH1 RC2: Classical Civilizations
Presentation
•
9th Grade

130 questions
2nd Semester Spanish I Review (includes Preterite)
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade