
Rate of reaction 1
Chemistry
5th Grade
NGSS covered
Used 628+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
13 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
What is the meaning of the rate of reaction?
Decrease in amount of product
Decrease in amount of product against time
Increase in amount of products against time
Increase in amount of reactants against time
Tags
NGSS.HS-PS1-5
2.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
Which unit is correct for the rate of reaction?
g mol-1
g min-1
mol dm-3
kJ mol-1
Tags
NGSS.HS-PS1-5
3.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
Which of the following has the lowest rate of reaction?
Combustion of ethanol
Fermentation of glucose
Oxidation of magnesium
Precipitation of silver chloride
Tags
NGSS.HS-PS1-5
4.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
Which reactions has the highest rate of reaction?
Rusting of water pipe
Photosynthesis in green plant
Burning of a small piece of charcoal in the air
Formation of stalactites and stalagmites
Tags
NGSS.HS-PS1-5
5.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Which process has the highest rate of reaction?
Rusting
Respiration
Combustion
Photosynthesis
Tags
NGSS.HS-PS1-5
6.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
The following equation represents a chemical equation.
CaCO3 + 2HCl --> CaCl2 + CO2 + H2O
Which graph shows the correct change in mass of reactant used in excess against time?


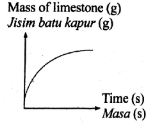
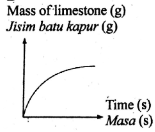
Tags
NGSS.HS-PS1-7
7.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
The rate of reaction for the decomposition of hydrogen peroxide decreases with time because
product of reaction decreases
temperature of hydrogen peroxide decreases
volume of hydrogen peroxide decreases
concentration of hydrogen peroxide decreases
Tags
NGSS.HS-PS1-5
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?

