Molecular Shapes Review
Authored by Nicole Fulmer
Chemistry
10th - 11th Grade
NGSS covered
Used 6+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
15 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the measure of a tetrahedral bond angle?
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the bond angle of H2O?
90
104.5
107.5
109.5
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the VSEPR theory used to predict?
4.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt

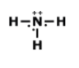
What 3-D VSEPR shape does this molecule exhibit?
linear
tetrahedral
trigonal pyramidal
trigonal planar
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
VSEPR stands for
Valid Shell Electricity Program Remedy
Valence Shell Electron Pair Repulsion
Velocity Sustaining Electron Proton Review
Valence Shell Electricity Proximal Repulsion
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What will repel one another according to VSEPR theory in this tutorial?
Electricity
Protons
Neutrons
Electrons
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a lone pair of electrons?
Electron pairs that are lonely
Electrons pairs that aren't bonded to another atom
Electrons that have paired together
When electricity has split atoms apart
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

17 questions
TEK 5C Periodic Trends
Quiz
•
9th - 12th Grade

15 questions
GEN CHEM 1 QUARTER 2 MODULE 2 POST TEST
Quiz
•
11th Grade

17 questions
8th grade science STAAR review 1
Quiz
•
KG - University

17 questions
Atom Scientists
Quiz
•
9th - 12th Grade

18 questions
Simple Lewis Dot Structures
Quiz
•
9th - 12th Grade

20 questions
Year 10 Chemical World
Quiz
•
8th - 11th Grade

16 questions
C10.1 GCSE Reactions of Alkenes AQA 8462
Quiz
•
9th - 11th Grade

15 questions
Naming Ionic Compounds
Quiz
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

20 questions
Chemistry: Classification of Matter
Quiz
•
10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade

20 questions
Stoichiometry Review
Quiz
•
10th Grade