
Quantum theory & Orbitals!
Authored by Luke Proctor
Chemistry
10th Grade
NGSS covered
Used 188+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
14 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt

Atoms are made up of three basic parts, which are -
Tags
NGSS.MS-PS1-1
2.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
Who proposed that the electrons could also have wavelike behaviors.
Bohr
Heisenberg
de Broglie
Tags
NGSS.HS-PS4-3
3.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
Since electrons are moving so fast, we can never know both their speed and position. Who said this?
Bohr
Heisenberg
Rutherford
4.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
Erwin Schrodinger proposed...
The Bohr model of atoms
The quantum mechanical model of atoms
No new discovery
That electrons are actually positive
5.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
The principle quantum number, n, represents the:
spin value
suborbital value
energy level
magnetic value
Tags
NGSS.HS-PS1-1
6.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
There are 4 different types of sublevels: s,p,d,f
Tags
NGSS.HS-PS1-1
7.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
Which shape represents the shape of an "s" orbital?
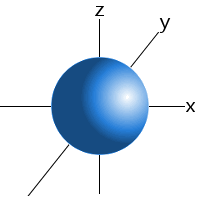
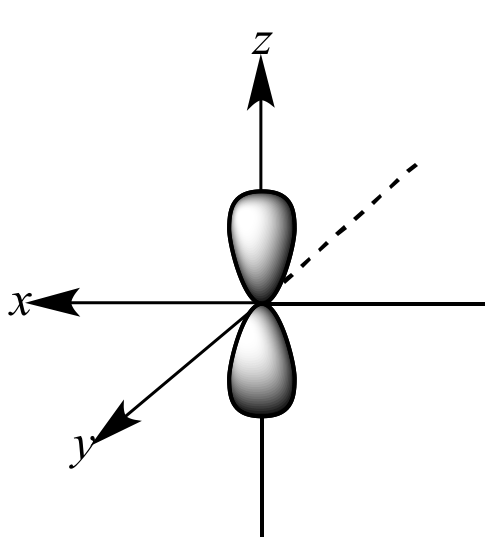
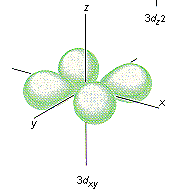
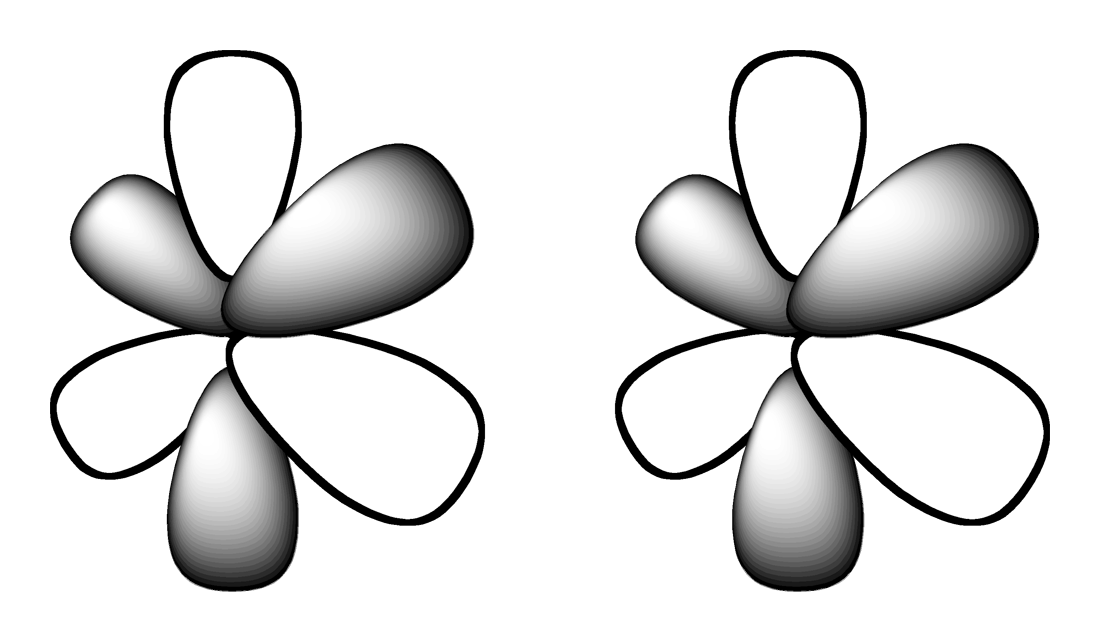
Tags
NGSS.HS-PS1-1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
