
Rate of Reactions
Authored by Adem Koc
Chemistry
9th - 11th Grade
NGSS covered
Used 45+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
25 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt
What is the meaning of the rate of reaction?
Decrease in amount of product
Decrease in amount of product against time
Increase in amount of products against time
Increase in amount of reactants against time
Tags
NGSS.HS-PS1-5
2.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt
The following equation represents a chemical equation.
CaCO3 + 2HCl --> CaCl2 + CO2 + H2O
Which graph shows the correct change in mass of reactant used in excess against time?


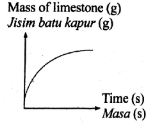
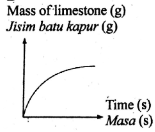
Tags
NGSS.HS-PS1-7
3.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt
The rate of reaction for the decomposition of hydrogen peroxide decreases with time because ...
concentration of water decreases
temperature of hydrogen peroxide decreases
volume of hydrogen peroxide decreases
concentration of hydrogen peroxide decreases
Tags
NGSS.HS-PS1-5
4.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt
In which of the chemical reactions can the rate be determined by measuring the change in the gas volume?
oxygen gas reacting with hydrogen gas
Sodium hydroxide solution with dilute hydrochloric acid
Silver nitrate solution with sodium chloride solution
Calcium carbonate with dilute hydrochloride acid
Tags
NGSS.HS-PS1-5
5.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt
The reaction between zinc, Zn and hydrochloric acid, HCl is represented by the following equation.
Zn + 2HCl --> ZnCl2 + H2
A student wants to determine the rate of reaction in a school laboratory. Which of the following methods is the most suitable?
Determine the change in temperature of the solution by time
Determine the change in the concentration of zinc chloride by time
Determine the volume of hydrogen gas given off by time
Determine the change in the concentration of hydrochloric acid by time
Tags
NGSS.HS-PS1-5
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
The following equation represents the reaction between calcium carbonate, CaCO3 and hydrochloric acid, HCl.
CaCO3 + 2HCl --> CaCl2 + CO2 + H2O
Which changes can be used to determine the rate of reaction?
I : mass of calcium carbonate per unit time
II : Volume of carbon dioxide released per unit time
III : Colour of solution per unit time
IV : Mass of precipitate produced per unit time
I and II
I and III
II and IV
III and IV
Tags
NGSS.HS-PS1-5
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt

Diagram shows a graph of volume of oxygen gas collected against time in the decomposition reaction of hydrogen peroxide when using manganese dioxide as catalyst.
Which point shows the highest rate of reaction?
P
Q
R
S
Tags
NGSS.HS-PS1-5
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
