
Practice Problems Unit 4 Lesson 1
Authored by Ryan McCluskey
Chemistry
11th Grade - University
NGSS covered
Used 7+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
25 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
What is the correct Lewis Dot Structure for ammonia NH3
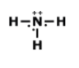
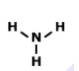
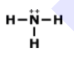
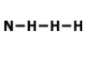
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
2.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
Which is the correct lewis structure for carbon dioxide?




Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
3.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

What is the formal charge on the oxygen atom at the top of this Lewis dot structure?
0
-1
+1
-3
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
4.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

What is the formal charge on the phosphorus atom in this Lewis dot structure?
0
-1
+1
-3
5.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

What is the formal charge on the bromine atom in this Lewis dot structure?
-1
+1
-2
+2
Tags
NGSS.HS-PS1-1
6.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
When forming bonds metals tend to __________.
Lose electrons forming positive ions
Lose electrons forming negative ions
Gain electrons forming positive ions
Gain electrons forming negative ions
7.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
How many total valence electrons are used in the lewis structure for H2O?
8
18
17
7
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?


