
Rates of Reaction
Authored by Tristen Greenwood
Chemistry
University
Used 2+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
15 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Why does a higher temperature increase the rate of a reaction?
it increases both the frequency and energy of particle collisions
it only increases the frequency of particle collisions
it only increases the energy of particle collisions
it reduces the activation energy of the reaction
2.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Grinding a effervescent tablet into powder increases the rate of reaction due to increased
concentration
surface area
temperature
reactants
3.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Adding a catalyst ___________ the activation energy.
Raises
Lowers
Doesn't affect
Inreases
4.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
The minimum amount of energy needed for colliding particles to react is called
Chemical Energy
Kinetic Energy
Activation Energy
Potential Energy
5.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Which graph shows the effect of increasing temperature on the rate of reaction of calcium carbonate with dilute hydrochloric acid?
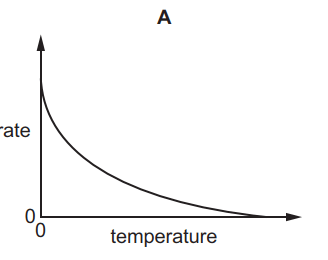
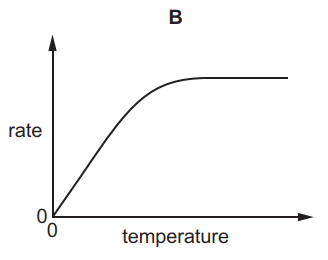
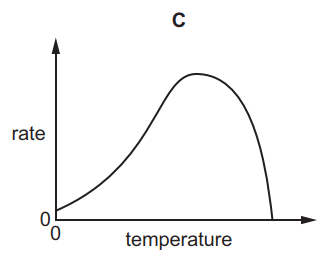
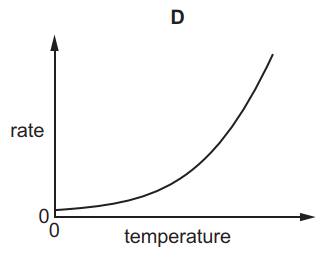
6.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Why does a catalyst increase the rate of reaction?
7.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Why don't all collisions between particles cause a reaction?
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?

