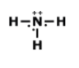
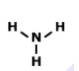
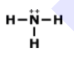
Unit 9 HW Lewis Structures
Quiz
•
Chemistry
•
11th Grade
•
Easy
Alison Klein
Used 5+ times
FREE Resource
Enhance your content in a minute
20 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to the octet rule most elements need _______ valence electrons.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Hydrogen needs _____ electrons in its valence shell to be stable.
3.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

Which of the following is the correct Lewis dot structure for the molecule fluorine (F2)?
4.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

What is the correct structure for BF3?
5.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
How many valence electrons are in Phosphorus?
6.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
How many electrons does each line indicate are shared?
7.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
When writing Lewis Structures, only __________ electrons are used.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

20 questions
Titration Method
Quiz
•
10th - 11th Grade

20 questions
Basics of Chemical Bonding
Quiz
•
10th - 11th Grade

25 questions
Naming and Balancing Chemical Reactions Review
Quiz
•
6th - 12th Grade

16 questions
Atomic Structure
Quiz
•
8th - 12th Grade

25 questions
GRHS Chemistry - Conservation of Matter Review
Quiz
•
10th - 12th Grade

15 questions
Properties of Acids & Bases
Quiz
•
8th - 12th Grade

18 questions
Balance Chemical Equation 3
Quiz
•
10th - 12th Grade

20 questions
Jadual Berkala Unsur
Quiz
•
KG - University
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

54 questions
Analyzing Line Graphs & Tables
Quiz
•
4th Grade

22 questions
fractions
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

15 questions
Equivalent Fractions
Quiz
•
4th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
electron configurations and orbital notation
Quiz
•
9th - 12th Grade

5 questions
Mole Conversions Made Easy
Interactive video
•
11th Grade

10 questions
16. Limiting Reagent/% Yield Practice
Quiz
•
11th Grade

20 questions
Metric Conversions
Quiz
•
11th Grade

10 questions
PERIODIC TRENDS
Quiz
•
11th Grade

14 questions
Reaction Types, Balancing, and Predicting Products
Quiz
•
9th - 12th Grade

8 questions
Empirical and Molecular Formulas
Lesson
•
9th - 12th Grade