
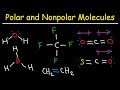
Polar and Non-Polar Molecules
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Easy
Emma Peterson
Used 12+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first category of non-polar molecules mentioned in the video?
Molecules with hydrogen bonding
Molecules made up of one element
Molecules with asymmetry
Molecules with high electronegativity difference
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is a non-polar molecule made up of only carbon and hydrogen?
Hydrogen fluoride (HF)
Ammonia (NH3)
Methane (CH4)
Water (H2O)
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What characteristic makes carbon tetrafluoride (CF4) a non-polar molecule?
Symmetry of the molecule
Asymmetry of the molecule
High electronegativity difference
Presence of hydrogen bonding
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If the electronegativity difference (EN difference) is less than 0.5, the molecule is generally:
Ionic
Non-polar
Polar
Metallic
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following molecules is non-polar due to a small EN difference?
Iodine monobromide (IBr)
Methanol (CH3OH)
Water (H2O)
Ammonia (NH3)
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bonding makes a molecule polar?
Metallic bonding
Hydrogen bonding
Van der Waals forces
Ionic bonding
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following molecules is polar due to hydrogen bonding?
Methane (CH4)
Carbon dioxide (CO2)
Oxygen (O2)
Ammonia (NH3)
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
