
Understanding Chemical Reactions: Systems and Surroundings
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Easy
Ethan Morris
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
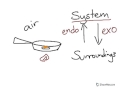
What is considered the 'system' in a chemical reaction?
The surroundings
The specific part being observed
The equipment used
The entire laboratory
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In an exothermic reaction, where does the energy flow?
From the surroundings to the system
Within the surroundings only
From the system to the surroundings
Within the system only
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following describes an endothermic reaction?
Energy is released to the surroundings
Energy is created within the system
No energy change occurs
Energy is absorbed from the surroundings
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the example of cooking an egg, what role does the egg play?
The catalyst
The energy source
The system
The surroundings
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When cooking an egg, what type of reaction is occurring?
Catalytic
Neutral
Endothermic
Exothermic
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the cooking example, what provides energy to the egg?
The handle
The fire
The air
The pan
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when the fire is considered the system?
It becomes endothermic
It remains unchanged
It releases energy to the surroundings
It absorbs energy from the egg
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
