
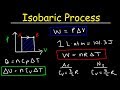
Thermodynamics and Gas Laws
Interactive Video
•
Physics, Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Olivia Brooks
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formula used to calculate the work done by a gas during an isobaric process?
W = nRT
W = PΔV
W = PV
W = nCvΔT
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In an isobaric process, if a gas expands, what is the sign of the work done?
Positive
Undefined
Zero
Negative
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which law relates the volume and temperature of a gas at constant pressure?
Boyle's Law
Charles's Law
Avogadro's Law
Dalton's Law
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the value of the gas constant R when using liters and atm?
8.3145 J/mol·K
0.08206 L·atm/mol·K
1.013 L·atm/mol·K
22.4 L/mol
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the temperature change when the volume of a gas doubles at constant pressure?
It triples
It doubles
It remains the same
It halves
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar heat capacity at constant pressure (Cp) for a monoatomic gas?
9/2 R
5/2 R
3/2 R
7/2 R
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following gases is considered monoatomic?
Carbon Dioxide
Helium
Nitrogen
Oxygen
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
