
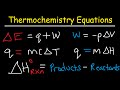
Thermochemistry Concepts and Calculations
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Medium
Emma Peterson
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the sign of Q when heat is released from a system?
Positive
Undefined
Negative
Zero
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If a gas expands from 2 L to 3 L at a pressure of 5 ATM, what is the sign of the work done by the system?
Zero
Negative
Undefined
Positive
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How much energy is required to heat 50 grams of water from 25°C to 75°C?
20,920 J
2,615 J
5,230 J
10,460 J
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the heat of fusion for water in kJ per mole?
4.184 kJ
6 kJ
18 kJ
12 kJ
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a combustion reaction, if 64 grams of oxygen reacts, how many kJ of heat energy will be released?
1,200 kJ
2,400 kJ
960 kJ
480 kJ
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the enthalpy change when 3600 kJ of heat energy is released in a reaction?
99 grams of CO2
792 grams of CO2
198 grams of CO2
396 grams of CO2
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the enthalpy of formation for CO2?
0 kJ/mol
-393 kJ/mol
-286 kJ/mol
-785 kJ/mol
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
