
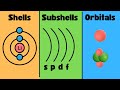
Understanding Energy Levels, Sublevels, and Orbitals
Interactive Video
•
1st Grade - University
•
Practice Problem
•
Medium
Aiden Montgomery
Used 31+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the principal quantum number associated with the first energy level?
2
1
3
4
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many electrons can the second energy level hold in total?
32
18
8
2
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which energy level can hold up to 18 electrons?
Level 3
Level 4
Level 2
Level 1
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the formula used to calculate the maximum number of electrons in an energy level?
2n^2
n^3
2n
n^2
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which sublevel can hold a maximum of 10 electrons?
f
p
s
d
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the maximum number of electrons that the f sublevel can accommodate?
18
10
6
14
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many orbitals are present in the p sublevel?
7
5
3
1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
