
Understanding Bond Lengths and Hybridization
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Jackson Turner
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which type of bond is generally longer, single or triple?
Quadruple bond
Single bond
Triple bond
Double bond
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of hybrid orbitals are involved in the formation of a carbon-carbon single bond in ethane?
sp hybrid orbitals
sp2 hybrid orbitals
sp3 hybrid orbitals
p orbitals
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How much s character is present in an sp3 hybrid orbital?
75%
25%
50%
100%
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is a triple bond shorter than a single bond?
It has less s character
It has more p character
It has less p character
It has more s character
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When comparing elements in the same row, what factor is crucial for determining bond length?
Electronegativity
P character
S character
Atomic number
6.
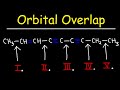
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which bond is formed from the overlap of an sp3 hybrid orbital and an sp2 hybrid orbital?
Bond number one
Bond number two
Bond number four
Bond number three
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which bond is the longest among the given options?
Bond number three
Bond number five
Bond number two
Bond number one
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
