
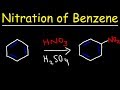
Nitration of Benzene Concepts
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Ethan Morris
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction is the nitration of benzene?
Addition reaction
Free radical substitution
Nucleophilic substitution
Electrophilic aromatic substitution
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which two acids are used to generate the electrophile in the nitration of benzene?
Sulfuric acid and acetic acid
Nitric acid and sulfuric acid
Nitric acid and hydrochloric acid
Hydrochloric acid and sulfuric acid
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of sulfuric acid in the generation of the electrophile?
It acts as a solvent
It acts as a catalyst
It provides a proton to make a better leaving group
It reacts with benzene
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the electrophile generated in the nitration of benzene?
Nitrate ion (NO3-)
Sulfate ion (SO4-)
Hydronium ion (H3O+)
Nitronium ion (NO2+)
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
During the reaction mechanism, what happens to one of the pi bonds in benzene?
It forms a new double bond
It breaks to form a carbocation
It forms a triple bond
It remains unchanged
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the intermediate formed when benzene reacts with the nitronium ion?
A radical
A carbocation
A neutral molecule
A carbanion
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is used to regenerate the aromatic ring in the nitration of benzene?
An acid
A base
A radical
A nucleophile
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
