
Metal-Ligand Interactions and Coordinate Bonds
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a key characteristic of a coordinate covalent bond?
Atoms share electrons equally.
One atom donates both electrons for the bond.
Both atoms donate one electron each.
Electrons are transferred completely.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a regular covalent bond between two bromine atoms, how many electrons does each atom contribute?
One electron
Two electrons
Three electrons
No electrons
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
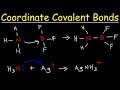
Why is the bond between nitrogen and boron in ammonia and boron trifluoride considered a coordinate covalent bond?
Both atoms share electrons equally.
Nitrogen donates both electrons for the bond.
Boron donates both electrons for the bond.
Electrons are transferred from boron to nitrogen.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What role does ammonia play in the reaction with boron trifluoride?
Neutral molecule
Electron acceptor
Lewis base
Lewis acid
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the metal-ligand interaction example, what is the charge of the silver cation?
Variable
Positive
Neutral
Negative
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the coordination number in the complex ion formed with ammonia and silver?
One
Two
Three
Four
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of interaction is involved in forming a complex ion with a metal and ligands?
Van der Waals forces
Hydrogen bonding
Metal-ligand interaction
Ionic interaction
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
