
Chemical Reaction Kinetics Concepts
Interactive Video
•
Chemistry, Science
•
10th Grade - University
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
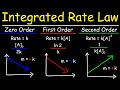
For a zero-order reaction, what happens to the rate if the concentration of reactant A is doubled?
The rate doubles.
The rate triples.
The rate remains unchanged.
The rate quadruples.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What are the units of the rate constant k for a first-order reaction?
M to the first power times T to the negative one
M to the zero power times T to the negative one
M to the negative two times T to the negative one
M to the negative one times T to the negative one
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which statement is true about the half-life of a first-order reaction?
It depends on the initial concentration.
It is independent of the initial concentration.
It decreases with the initial concentration.
It increases with the initial concentration.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the integrated rate law expression for a first-order reaction?
Final concentration = -kt + initial concentration
ln(final concentration) = -kt + ln(initial concentration)
1/final concentration = kt + 1/initial concentration
Final concentration = kt + initial concentration
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
For a second-order reaction, what plot gives a straight line?
1/concentration vs. time
Rate vs. concentration
ln(concentration) vs. time
Concentration vs. time
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If a reaction is third order overall, what are the units of the rate constant k?
M to the first power times T to the negative one
M to the zero power times T to the negative one
M to the negative two times T to the negative one
M to the negative three times T to the negative one
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which plot corresponds to a zero-order reaction?
Concentration vs. time with a positive slope
Concentration vs. time with a negative slope
ln(concentration) vs. time with a negative slope
1/concentration vs. time with a positive slope
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
