
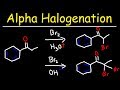
Alpha Halogenation of Ketones
Interactive Video
•
Chemistry, Science
•
10th - 12th Grade
•
Practice Problem
•
Hard
Ethan Morris
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the major product when acetone reacts with bromine under acidic conditions?
Acetone with one bromine atom replacing an alpha hydrogen
Acetone with two bromine atoms replacing alpha hydrogens
Acetone with no bromine atoms
Acetone with a hydroxyl group
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in the mechanism of alpha halogenation under acidic conditions?
Protonation of the ketone by hydronium ion
Direct reaction with bromine
Formation of the enolate ion
Deprotonation of the alpha hydrogen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the acidic mechanism, what form does the ketone convert into before reacting with bromine?
Radical form
Enol form
Carbanion form
Enolate form
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the intermediate formed during the basic mechanism of alpha halogenation?
Enolate ion
Enol
Carbocation
Radical
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the enolate ion react with bromine in the basic mechanism?
By attacking the bromine molecule
By losing a proton
By forming a pi bond
By forming a radical
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the practice problem under acidic conditions, how many alpha hydrogens are replaced?
One
Two
None
All
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the major product when a ketone reacts with excess bromine under acidic conditions?
Ketone with one bromine atom
Ketone with two bromine atoms
Ketone with no bromine atoms
Ketone with a hydroxyl group
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
