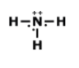
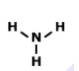
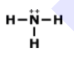
Lewis Structures for Covalent Compounds
Authored by Valerie Trevino
Chemistry
11th Grade
NGSS covered
Used 117+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
20 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Which electrons are involved in the formation of a chemical bond?
dipoles
s electrons
Lewis Structures
valence electrons
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
2.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
How many electrons are involved in a triple bond?
2
4
6
8
3.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
How many electrons are involved in a double bond?
2
4
6
8
4.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
How many electrons are involved in a single bond?
2
4
6
8
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
5.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
In MOST compounds, atoms form bonds so that the number of valence electrons of each atom is ____.
2
6
8
10
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
6.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
The electrical attraction of an atom for the electrons is called --
electron affinity
electronegativity
resonance
ionization energy
Tags
NGSS.HS-PS1-1
7.
MULTIPLE CHOICE QUESTION
2 mins • 1 pt
To draw a Lewis structure, we need to find the --
number of valence electrons in each atom.
atomic mass of each atom.
bond length of each atom.
ionization energy of each atom.
Tags
NGSS.HS-PS1-1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?