VSEPR and Polarity Lesson
Authored by Charles Martinez
Chemistry
9th - 12th Grade
NGSS covered

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
15 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

CO2 has polar bonds but is a NON POLAR molecule. Why?
it has an asymmetrical shape
bond polarity or dipole moment between C and O atoms cancel
there is net dipole moment between C and O atoms in molecule
bond polarity does not exist between C+ and O- atoms
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Does the following reference Polar, Nonpolar, or both:
"electronegativity values are the same"?
Polar
Nonpolar
Both
3.
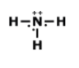
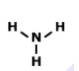
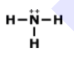
MULTIPLE CHOICE QUESTION
45 sec • 1 pt

Is this molecule polar or nonpolar?
polar
nonpolar
Tags
NGSS.HS-PS1-1
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to VSEPR, molecules adjust their shapes to keep which of the following as far away as possible?
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a polar bond, the more electronegative element will assume a partial ________ charge.
6.
MULTIPLE CHOICE QUESTION
45 sec • 1 pt
Type of bond between Br & Br
Ionic
Polar Covalent
Nonpolar Covalent
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which molecule contains bonds with a GREATER polarity?
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?