Introduction to Chemical Bonding
Authored by Charles Martinez
Chemistry
9th Grade
NGSS covered
Used 4+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
15 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Typically, atoms are more stable when they are
bonded together
apart from each other
2.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Why do atoms bond?
They typically don't bond
To add or take away energy levels
To have a full valance shell.
To have a full inner shell
Tags
NGSS.HS-PS1-2
NGSS.HS-PS1-1
3.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Ionic bonding is between a
nonmetal and nonmetal
metal and nonmetal
metal and metal
Depends on the situation
Tags
NGSS.HS-PS1-3
4.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt
Covalent bonding is between a
nonmetal and nonmetal
metal and nonmetal
metal and metal
It depends on the situation
5.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt

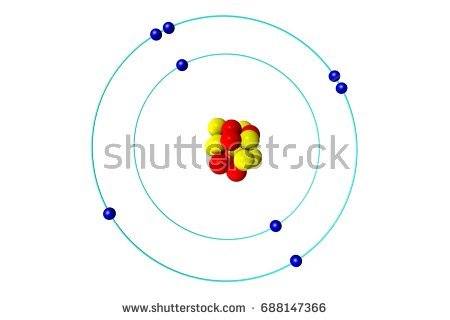
Which bond does this picture best represent?
Metallic bond
ionic bond
covalent bond
James Bond
6.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt
What do positive ions tend to do?
lose electrons
gain electrons
lose protons
gain protons
7.
MULTIPLE CHOICE QUESTION
20 sec • 1 pt
What happens when magnesium loses 2 electrons?
It stabilizes to a net charge of 0
It turns into an atom
It becomes negatively charged
It becomes positively charged
Tags
NGSS.HS-PS1-2
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

19 questions
Quimica 8º
Quiz
•
1st - 10th Grade

20 questions
Ionic and Covalent Bonding Y10
Quiz
•
7th - 11th Grade

20 questions
Símbolos Químicos
Quiz
•
1st - 12th Grade

16 questions
Electricity Quiz
Quiz
•
KG - University

15 questions
Atoms, Isotopes, and bonds
Quiz
•
9th - 12th Grade

20 questions
Nomenclature Quiz
Quiz
•
9th - 12th Grade

13 questions
Elements and the Periodic Table
Quiz
•
9th Grade

10 questions
Hydrocarbons
Quiz
•
9th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

54 questions
Analyzing Line Graphs & Tables
Quiz
•
4th Grade

22 questions
fractions
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

15 questions
Equivalent Fractions
Quiz
•
4th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

10 questions
Formative 3BC: Ionic v Covalent Bonds
Quiz
•
9th Grade

10 questions
Exploring Stoichiometry Concepts
Interactive video
•
6th - 10th Grade

20 questions
Mixed Bonding Naming
Quiz
•
9th Grade

20 questions
Chemical Reactions
Quiz
•
9th Grade

20 questions
electron configurations and orbital notation
Quiz
•
9th - 12th Grade

14 questions
Reaction Types, Balancing, and Predicting Products
Quiz
•
9th - 12th Grade

8 questions
Empirical and Molecular Formulas
Lesson
•
9th - 12th Grade