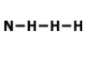Drawing Lewis Dot Structures
Authored by Lisa Thompson
Science
10th Grade
NGSS covered
Used 1+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
15 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
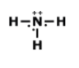
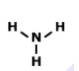
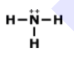
How many electrons should Nitrogen have around its Lewis dot model?
1
3
4
5
2.
REORDER QUESTION
1 min • 1 pt
Reorder the following steps for drawing lewis structures
Add remaining electrons as Lone pairs (terminal atoms first).
If any atoms dont have an Octet, use electrons to create double bonds.
The Valence electrons
charge - add/subtract if there is a charge
Check each atom has the same number of electrons as it started with (formal charge)
Place the lower Electronegative atom in the centre and draw in bonds
3.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

Which of the following is the correct Lewis structure for the compound PBr3?
structure A
structure B
structure C
structure D
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
4.
DRAW QUESTION
1 min • 1 pt
Draw the lewis structure for CH3NH2

Tags
NGSS.HS-PS1-1
5.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

This could be the dot diagram of
Mg
Cl
C
O
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
6.
MULTIPLE CHOICE QUESTION
1 min • 12 pts
Each line in a Lewis Structure represents __________ electron(s).
3
2
1
4
7.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Which Lewis Dot Model drawing correctly shows an O2 molecule?




Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?