
Chemical Reactions Amplify
Authored by Lisa Thompson
Science
7th Grade
NGSS covered

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
15 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

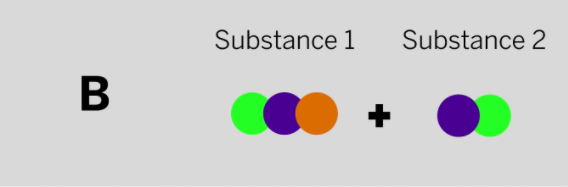
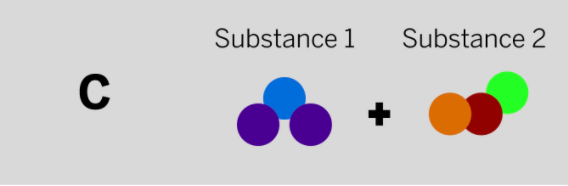
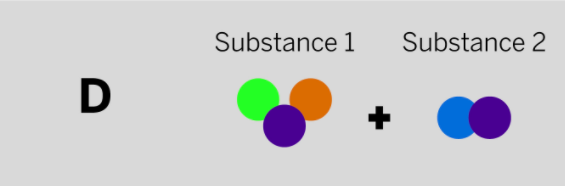
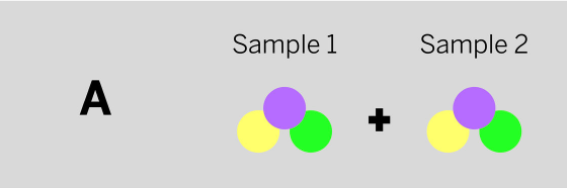
Jamie works at a company that makes cleaning chemicals. She is trying to make a chemical that smells like flowers. She took two samples that were gases at room temperature and mixed them in a sealed container. The diagram above shows the repeating groups of atoms that make up the two starting substances.
After mixing, Jamie found two substances that smelled like flowers in the sealed container. (Nothing had escaped.) Which of the diagrams to the left shows the repeating groups of atoms that make up the ending substances?

Tags
NGSS.MS-PS1-2
NGSS.MS-PS1-5
NGSS.MS-PS1-1
2.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

A chemist mixed two samples together: a gray liquid that boils at 103°C and a colorless gas that boils at 19°C. She analyzed the results and found two ending substances. One of the ending substances was a pink liquid. This ending substance is made up of the repeating group of atoms shown above. Which of the diagrams to the left shows the repeating groups of atoms that make up the samples the chemist mixed together?

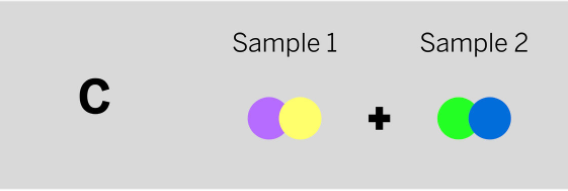

Tags
NGSS.MS-PS1-2
NGSS.MS-PS1-5
NGSS.MS-PS1-1
3.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

Patrik works at a company that makes paper and needs a chemical to make the paper brighter. He mixed two colorless substances together in a sealed container. The diagram above shows the repeating groups of atoms that make up the two starting substances.
After mixing, Patrik found two white substances in the sealed container. (Nothing had escaped.) Which of the diagrams to the left shows the repeating groups of atoms that make up the ending substances?
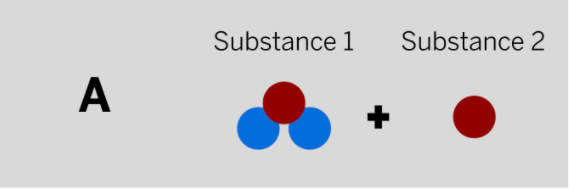
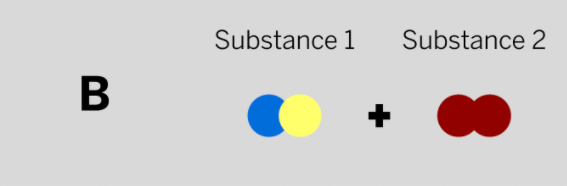
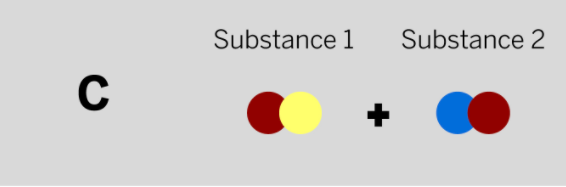
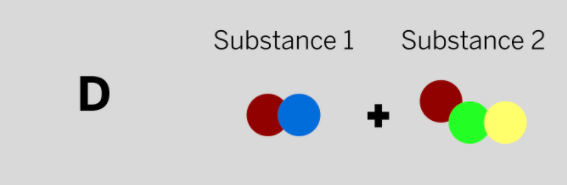
Tags
NGSS.MS-PS1-2
NGSS.MS-PS1-5
NGSS.MS-PS1-1
4.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

Serena mixed two samples together: a gray solid that smells like flowers and a gray solid that smells like bleach. She analyzed the results and found two ending substances. One of the ending substances was a white solid. This ending substance is made up of the repeating group of atoms shown above. Which of the diagrams to the left shows the repeating groups of atoms that make up the samples Serena mixed together?




Tags
NGSS.MS-PS1-2
NGSS.MS-PS1-5
NGSS.MS-PS1-1
5.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

Marisol mixed two samples together: a brown liquid with no smell and a colorless gas that smells irritating. She analyzed the results and found two ending substances. One of the ending substances smelled sweet. This ending substance is made up of the repeating group of atoms shown above. Which of the diagrams to the left shows the repeating groups of atoms that make up the samples Marisol mixed together?
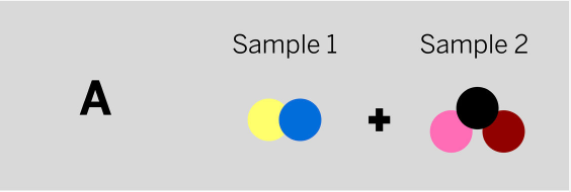
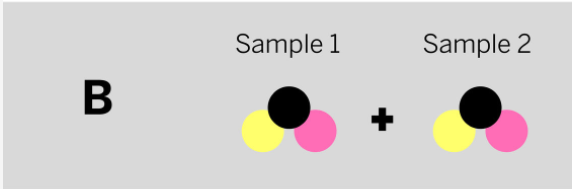

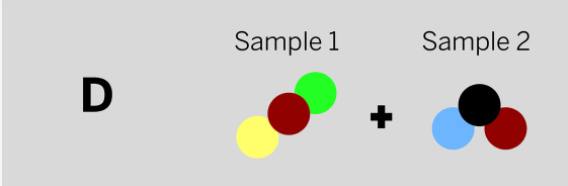
Tags
NGSS.MS-PS1-2
NGSS.MS-PS1-5
NGSS.MS-PS1-1
6.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

Andres is a chemist working at a company that makes ink. To make a new kind of ink, Andres mixed two liquid substances together in a sealed container. The diagram above shows the repeating groups of atoms that make up the two starting substances.
After mixing, Andres found two solid substances in the sealed container. (Nothing had escaped.) Which of the diagrams to the left shows the repeating groups of atoms that make up the ending substances?




Tags
NGSS.MS-PS1-2
NGSS.MS-PS1-5
NGSS.MS-PS1-1
7.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

Samira watched a demonstration in which a scientist combined two substances in a sealed container and analyzed the results. The ending substances did not look the same as the starting substances. Samira was given a diagram with the repeating groups of atoms that make up the starting substances, plus information about the properties of the starting and ending substances. She created a model of the repeating groups of atoms that might make up the two ending substances.
Samira's model correctly demonstrates how the properties changed with the rearrangement of the atoms. However not all atoms are accounted for. There is a missing reactant.
Samira's model is correct because the properties are different and the atoms are rearranged.
Samira's model is incorrect because not all atoms are accounted for
Samira's model correctly demonstrates how the properties changed with the rearrangement of the atoms. However not all atoms are accounted for. There is a missing product.
Tags
NGSS.MS-PS1-2
NGSS.MS-PS1-5
NGSS.MS-PS1-1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?







