
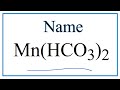
Naming and Charges of Polyatomic Ions
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of metal is manganese in the compound MnH(CO3)2?
Transition metal
Alkaline earth metal
Noble gas
Alkali metal
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in naming the compound MnH(CO3)2?
Calculate the net charge
Write the name of the metal as it appears on the periodic table
Determine the charge of the metal
Identify the polyatomic ion
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the polyatomic ion present in MnH(CO3)2?
Nitrate
Bicarbonate
Sulfate
Carbonate
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is another name for the bicarbonate ion?
Sulfate ion
Hydrogen carbonate ion
Carbonate ion
Nitrate ion
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it necessary to indicate the charge of manganese in the compound name?
Because manganese is a transition metal
Because manganese is a non-metal
To differentiate it from other metals
To indicate the number of polyatomic ions
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the bicarbonate ion in MnH(CO3)2?
1+
2+
1-
2-
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you represent the charge of manganese in the compound name?
Using a subscript
With a suffix
With a prefix
By a Roman numeral in parentheses
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
