
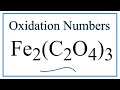
Oxidation Numbers and Charges
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Aiden Montgomery
FREE Resource
Read more
9 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the overall charge of the compound Iron(III) Oxalate?
Positive
Variable
Negative
Neutral
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge of the oxalate ion (C2O4)?
1+
3+
0
2-
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do the oxidation numbers in an ion relate to the ion's charge?
They are always negative.
They add up to the ion's charge.
They add up to zero.
They are always positive.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
If there are three oxalate ions, what is the total oxidation number for these ions?
-3
0
-6
+6
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What oxidation number must each iron atom have to balance the compound?
+4
+6
+3
+2
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the sign of an oxidation number written compared to an ionic charge?
After the number for both.
After the number for oxidation numbers, before for ionic charges.
Before the number for both.
Before the number for oxidation numbers, after for ionic charges.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the oxidation number of iron in Iron(III) Oxalate?
+2
+3
+4
+5
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

6 questions
VOICED : Gimnasia rumana en crisis lejos de viejas glorias
Interactive video
•
9th - 10th Grade

11 questions
How to say THIS, THAT, THESE & THOSE in French
Interactive video
•
10th - 12th Grade

6 questions
CLEAN: Chelsea lose star performers to Africa Cup of Nations
Interactive video
•
9th - 10th Grade

6 questions
Coronavirus Model Predicts U.S. Death Toll Of Over 200,000 By October
Interactive video
•
9th - 12th Grade

6 questions
CLEAN : Liberia war crimes trial resumes in Finland
Interactive video
•
9th - 12th Grade

6 questions
CLEAN : Charlie Hebdo victims added to journalist memorial
Interactive video
•
9th - 12th Grade
Popular Resources on Wayground

7 questions
History of Valentine's Day
Interactive video
•
4th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

15 questions
Valentine's Day Trivia
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

19 questions
Stoichiometry, Limiting Reactants, and Percent Yield
Quiz
•
10th Grade

10 questions
Formative 3BD: Ionic Bonds
Quiz
•
9th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

10 questions
Identifying types of reactions
Quiz
•
9th - 12th Grade

20 questions
Periodic Trends
Quiz
•
10th Grade