
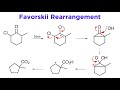
Favorskii Rearrangement Concepts
Interactive Video
•
Chemistry
•
11th - 12th Grade
•
Practice Problem
•
Hard
Liam Anderson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Who invented the Favorskii rearrangement?
Dmitri Mendeleev
Alexei Favorskii
Linus Pauling
Marie Curie
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the primary application of the Favorskii rearrangement in synthesis?
Ring expansion
Ring contraction
Oxidation
Polymerization
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the Favorskii rearrangement, what intermediate is formed after the enolate attacks the carbon bearing the chloro group?
Cyclopentanone
Cyclobutanone
Cyclohexanone
Cyclopropanone
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the cyclopropanone intermediate in the Favorskii rearrangement?
It is stable and can be isolated
It forms a stable dimer
It is attacked by hydroxide base
It undergoes a Diels-Alder reaction
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a non-symmetrical system, how does the ring open in the Favorskii rearrangement?
To yield a radical
To yield a symmetrical carbanion
To yield the less substituted carbanion
To yield the more substituted carbanion
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a key feature of the Favorskii rearrangement mechanism?
It only works with symmetrical ketones
It yields the same carboxylate from isomeric alpha-haloketones
It requires a metal catalyst
It yields different carboxylates from isomeric alpha-haloketones
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when anhydrous alkoxides are used as bases in the Favorskii rearrangement?
Alcohols are formed
Esters are formed
Aldehydes are formed
Carboxylate salts are formed
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
