
Carbonate Chemistry Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of ion is carbonate considered to be?
Monatomic ion
Polyatomic ion
Cation
Anion
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why are carbonates generally considered insoluble in water?
They form strong covalent bonds.
They are nonpolar molecules.
They tend to form precipitates.
They react violently with water.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does 'ss' indicate in a solubility table?
Strongly soluble
Slightly soluble
Solid state
Saturated solution
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of a solubility table in this context?
To measure pH levels
To calculate molarity
To determine solubility
To predict reaction speed
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
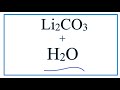
When lithium carbonate dissolves in water, what ions are formed?
Li+ and CO2 3-
Li- and CO3+
Li2+ and CO3-
Li+ and CO3 2-
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is a coefficient of 2 placed in front of Li in the equation?
To balance the charge
To show complete dissolution
To account for two lithium ions
To indicate a solid state
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of the subscript '2' in Li2CO3?
It means the compound is highly reactive.
It shows the compound is a gas.
It suggests the compound is a liquid.
It indicates two lithium atoms are present.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
