
Stoichiometry Concepts and Calculations
Interactive Video
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Hard
Jackson Turner
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the central unit used in stoichiometry for conversions?
Moles
Liters
Particles
Grams
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
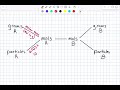
What tool does the teacher suggest using to simplify stoichiometry?
Textbook
Calculator
Road map
Laboratory equipment
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is used to convert grams to moles?
Balanced equation
Coefficient ratio
Avogadro's number
Molar mass
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What number is essential for converting particles to moles and vice versa?
Molecular weight
Density
Avogadro's number
Atomic number
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What must be memorized to facilitate conversions in stoichiometry?
Periodic table
Chemical names
Laboratory procedures
Conversion roadmap
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass used for in stoichiometry?
To balance equations
To convert grams to moles
To measure temperature
To identify substances
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the significance of a balanced equation in stoichiometry?
It reduces the amount of reactants used
It speeds up chemical reactions
It is necessary for correct mole ratios
It ensures accurate temperature readings
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
