
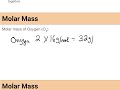
Exploring Moles and Molar Mass Concepts
Interactive Video
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Easy
Ethan Morris
Used 1+ times
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the numerical value that defines one mole?
100
1 x 10^12
12
6.02 x 10^23
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What common unit is used to compare with a mole for better understanding?
A pair
A dozen
A score
A hundred
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does the molar mass of a substance represent?
The mass of one mole of the substance
The volume of one mole of the substance
The number of particles in a mole
The density of the substance
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you find the molar mass of a compound?
Multiply the atomic number by Avogadro's number
Use the density and volume of the compound
Divide the molecular weight by Avogadro's number
Add the mass of each element from the periodic table
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of oxygen if it is diatomic and each atom weighs approximately 16 g/mol?
64 g/mol
8 g/mol
32 g/mol
16 g/mol
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total molar mass of methane (CH4)?
28 g/mol
16 g/mol
12 g/mol
4 g/mol
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many atoms of hydrogen are present in a molecule of methane?
1
2
4
8
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
