
Thermochemistry Concepts and Applications
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Aiden Montgomery
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
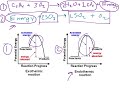
What is the main focus of thermochemical equations?
The color change in reactions
The energy absorbed or released during reactions
The speed of chemical reactions
The physical state of reactants and products
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In an exothermic reaction, where is the energy listed in the chemical equation?
It is not listed
In the middle of the equation
On the product side
On the reactant side
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is activation energy?
The energy stored in the reactants
The energy absorbed by the products
The energy released during a reaction
The energy required to start a reaction
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the change in enthalpy (ΔH) of a reaction?
By observing the color change
By counting the number of bonds formed
By calculating the difference in energy between reactants and products
By measuring the temperature change
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does a positive ΔH indicate about a reaction?
It is exothermic
It is endothermic
It releases energy
It does not involve energy change
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to bonds during an endothermic process?
Bonds are both formed and broken
Bonds are broken
Bonds are formed
No bonds are involved
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which process releases energy: bond breaking or bond forming?
Bond breaking
Both release energy
Neither release energy
Bond forming
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
