
Exploring Endothermic and Exothermic Reactions
Interactive Video
•
Chemistry
•
6th - 10th Grade
•
Practice Problem
•
Hard
Aiden Montgomery
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction absorbs energy?
Endothermic
Exothermic
Both
Neither
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which reaction requires energy input to proceed?
Exothermic
Endothermic
Both
Neither
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
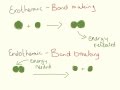
Which reaction is associated with bond making?
Endothermic
Neither
Exothermic
Both
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a common example of an exothermic reaction?
Photosynthesis
Dissolving salt in water
Burning
Melting ice
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction is thermal decomposition?
Endothermic
Exothermic
Neither
Both
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is likely to appear on the exam related to reactions?
A historical context question
A complex math question
A practical lab question
A simple definition question
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is an example of an endothermic reaction?
Respiration
Thermal decomposition
Neutralization
Combustion
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
