
Chemical Reactions and Stoichiometry
Interactive Video
•
Chemistry, Mathematics
•
10th - 12th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
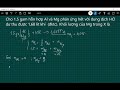
What is the total mass of the mixture of aluminum and magnesium used in the reaction?
2.5 grams
1.5 grams
2.0 grams
1.0 gram
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which gas is produced when aluminum and magnesium react with hydrochloric acid?
Carbon Dioxide
Oxygen
Nitrogen
Hydrogen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the valency of aluminum in the reaction?
1
2
3
4
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the valency of magnesium different from that of aluminum?
Magnesium has a valency of 4
Magnesium has a valency of 1
Magnesium has a valency of 2
Magnesium has a valency of 3
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the volume of hydrogen gas produced in the reaction?
1.68 liters
2.58 liters
2.68 liters
1.58 liters
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do you calculate the number of moles of hydrogen gas?
Volume divided by 24
Volume multiplied by 22.4
Volume divided by 22.4
Volume multiplied by 24
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in setting up the system of equations for this problem?
Calculate the mass of hydrogen
Determine the moles of aluminum
Find the volume of magnesium
Establish the relationship between moles and mass
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
