
Understanding Bond Energy
Interactive Video
•
Chemistry, Science
•
9th - 12th Grade
•
Practice Problem
•
Hard
Jackson Turner
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
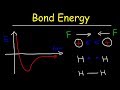
What happens to the energy of a system when two atoms are infinitely far apart?
The energy becomes positive.
The energy becomes zero.
The energy becomes infinite.
The energy becomes negative.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
At what point is the bond length of a molecule determined?
When the energy is positive.
When the energy is zero.
When the energy is at its minimum.
When the energy is at its maximum.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to the energy of the system when atoms are extremely close to each other?
The energy decreases and becomes negative.
The energy becomes zero.
The energy remains constant.
The energy increases and becomes positive.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does the energy increase when atoms are very close to each other?
Due to electron-proton repulsion.
Due to proton-proton and electron-electron repulsion.
Due to electron-electron attraction.
Due to proton-proton attraction.
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does a positive energy value indicate about the forces between atoms?
The forces are balanced.
The forces are neutral.
The forces are repulsive.
The forces are attractive.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does a negative energy value indicate about the forces between atoms?
The forces are attractive.
The forces are repulsive.
The forces are neutral.
The forces are balanced.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the hydrogen atom example, what type of bond is formed between the two hydrogen atoms?
Metallic bond
Non-polar covalent bond
Polar covalent bond
Ionic bond
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
